Example:
HOCl > HOBr > HOI (as decreasing order of electronegativity is Cl > Br > I)
When the element X acquire additional oxygen, the electron pulling power by the group X further increases due to increase of –I inductive effect exerted by the additional oxygen atom.
Example:
HOCl < HOClO < HOClO2 < HOClO3
It is also to note the number of oxo group operation of the number of O - H bonds
Example:
H3PO3 is more acidic than that of H3PO4
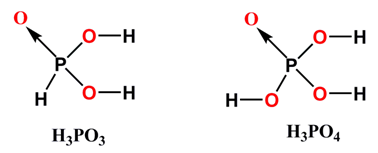
In case of H3PO3 one oxo group affect on two O - H bonds, while in H3PO4 one oxo group affect on three O - H bonds. Therefore, because of more –I inductive effect experienced by the O - H bonds present in H3PO3 , H3PO4 behaves as less acidic.
Acid base behavior of metal compounds:
Metal oxides are generally basic in nature. Few of them are amphoteric. In the periodic table oxides becomes more basic on moving from top to bottom of the table, and more acidic on going from left to right.
Example:
BeO is amphoteric, MgO, CaO, SrO, BaO are basic and basicity increases from BaO to MgO
Na2O, MgO are basic, Al2O3 is amphoteric, SiO2, P4O10 , SO3 , Cl2O7 are acidic and the acidity increases from SiO2 to Cl2O7
With an increase in charge/radious ratio the acidity of metal oxides increases.
Non aqueous solvents and ionic liquids:
Studies on non-aqueous system, particularly on liquid ammonia revealed that auto-ionization of liquid NH3 similar to water occurs.
![]()
Akin to substances producing H3O+ ions in water acts as acids in aqueous medium, substances that produce NH4+ ions in liquid ammonia medium acts as acids and it may also be expected to behave as an acid in that medium. Similarly, NH2− producing substance behaves as a base in liquid ammonia medium.
Acids in liquid ammonia medium: NH4Cl, NH4NO3 , (NH4)2SO4, etc.
Bases in liquid ammonia medium: KNH2 , NaNH2 , etc.
Similarly, SOCl2 behaves as an acid and CaSO3 behaves as a base in liquid SO2 solvent and their neutralization reaction can be expressed as
![]()
Acid–base behavior is also known to show by some other ionic pair (salts). e.g. In BrF3 , BrF2 AsF6 increases the concentration of the cation and hence the salt is defined as an acid in the solvent system.