Strength of Lewis acids and bases [Hard (H) and Soft (S) Acid (A) Base (B) Concept = HSAB Concept), 1963]:
Lewis defines acids are electrons acceptor and bases are electron donor. Therefore, the strength of acids and bases is determined by the nature of electron transfer in a particular reaction. Hence, the strength is dependent on a particular reaction. Accordingly, assignment of any single consistent criterion for acid-base strength becomes very difficult in the Lewis definition. However, a qualitative correlation between the various Lewis acids and Bases has been obtained by classifying the acids and bases in to two different groups, known as hard and soft.
In 1963, R. G. Pearson proposed that hard acids prefer to combine with hard bases and soft acids prefer to combine with soft bases. This is known as HSAB concept or theory.
The features distinguish Hard and Soft acids:

The features distinguish Hard and Soft bases:
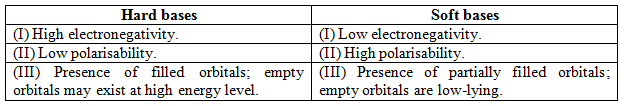
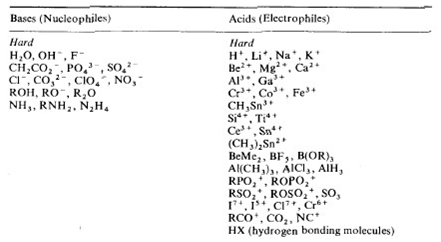
Table showing strength of HSAB, and border line case:
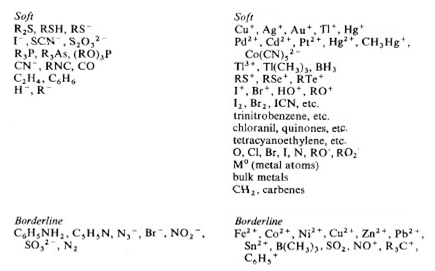
General trends in acid strength:
Protonic acids can be classified in two groups, (A) hydro-acids, and (B) oxo-acids.
(A) Hydro-acids: In hydro-acids proton(s) is(are) directly attached to some second element. In the periodic table the acidity of hydro-acids increases from left to right along a period and top to bottom along a group.
Example: HF > H2O > NH3
Here, on going from left to right in a period, electronegativity of the elements attached to proton increases, therefore, the proton-element bond become more polar. As the polarity of the bond increases, release of proton becomes easier. Hence, acidity increases.
Example: HI > HBr > HCl > HF
In this case, the size of the elements increases and electronegativity decreases on moving from top to bottom in a group. Here, size is the dominating factor over electronegativity. As size increases, the extent of overlap between small proton and big elements becomes low and proton-element bond becomes weak. Therefore, the release of proton becomes easier as the size of element increases, consequently, acidity increases.
(B) Strength of oxy-acids: In case of oxo acid of type X - O - H, the acidity of the acid decreases with lower the position of X in the periodical table. As we go down along a column in a periodical table, the electronegativity decreases. Hence, the electron pulling effect by the X through s -induction decreases. Therefore, the polarity of the H-O bond decreases along a column on going from top to bottom.