Conjugate acid-base pair:
Two chemical species, which are inter-convertible to each other by means of a proton, are known as conjugate acid-base pairs.
Example:
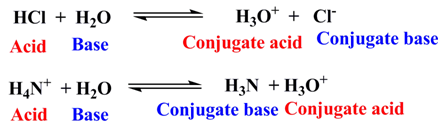
A species, which has high tendency to give up proton(s) in a medium, will behave as a strong acid. The corresponding conjugate base, then, must be weak in nature, i . e . its tendency for accepting proton(s) will be very low. The strength of an acid-species is also dependent to the stability of the corresponding conjugate base. The higher stability of the conjugate base favors the deprotonation process by the easy release of proton.
Example:
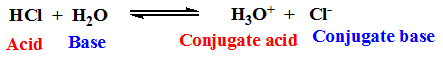
Similarly, a conjugate acid of a strong base should be weak in nature.
Example:
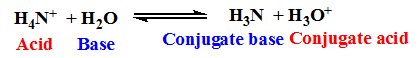
Merits and demerits:
Merits:
Acidic-basic properties of substances can be explained in any protonic solvent such as liquid ammonia, sulphuric acid, etc.
With the help of this theory the strength of acids and bases can be calculated. Further more, the reason that tunes the strength of acidic and basic properties (conjugate acid-base theory) can be understood.
Demerit:
In this theory, the acid-base properties are determined solely by proton exchange parameter. Therefore, some other substances that do not contain any proton but have inherent acidic or basic properties, e . g . BF3 , I2 , AlCl3 , etc cannot be explained with the help of the theory.