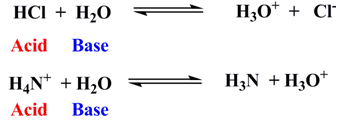
The Arrhenius Theory (1880-1890):
An acid is any hydrogen-containing compound that provides hydrogen ion in aqueous medium. Similarly, according to the theory, a base is any compound that gives hydroxyl ion in aqueous medium.
Example:
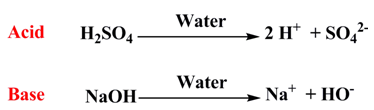
Merit:
- With the help of this theory, the strength of acids and bases could be explained in terms of ionization equilibrium:
- The existence of bare H+ in aqueous medium was questioned. The size of proton is extremely small and therefore, it was proposed that it combined with water molecule and exists as H3O+ (hydronium) ion.
Franklin Theory (1905):
This theory concerns the acidic and basic properties of a substance in non-aqueous medium, mainly, in ammonium.
According to this theory,
An acid is a solute that gives the cationic character of the solvent, and a base is a solute that provides anionic character of the solvent.
Similar to aqueous medium,
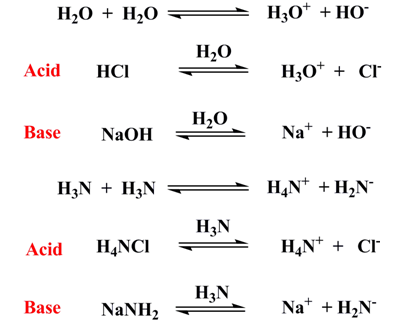
Merit and demerits:
Arrhenius theory was limited to aqueous medium, while, with the help of this theory acidic and basic properties of some substances in non-aqueous medium can be explained.
This definition also failed to define inherent acid-base character of a substance. Isotope labeling experiments showed that there was no dissociation and association of substance and solvent, hence, autoionization of solvents was questioned.
The Protonic Theory (Brønsted-Lowry concept, 1923):
Any species that tends to give up a proton is an acid, and any species that tends to accept a proton is a base.
Examples: