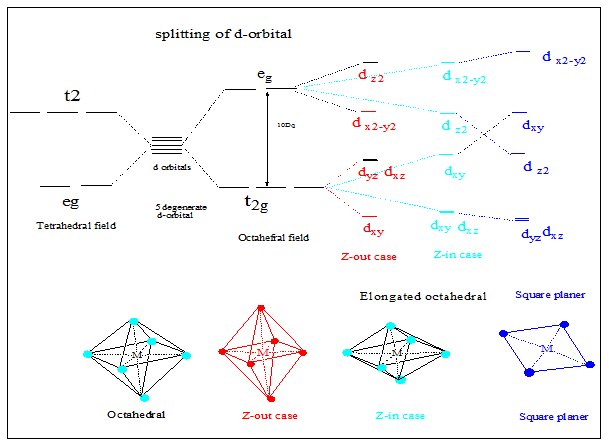
Splitting of d -orbitals in tetrahedral complexes:
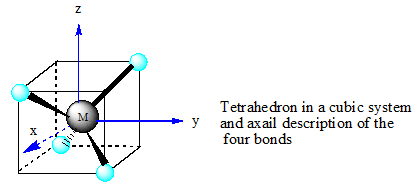
The tetrahedral structure suggest that orbital lobes in between two axes interact with the ligand electron pair and hence those orbitals (dxy, dxz and dyz) experience stronger repulsion and energy of those orbitals rise where as no ligand along any axis help to lower down the energy of orbital having lobes along any axis(dx2−y2 and dz2 orbitals).
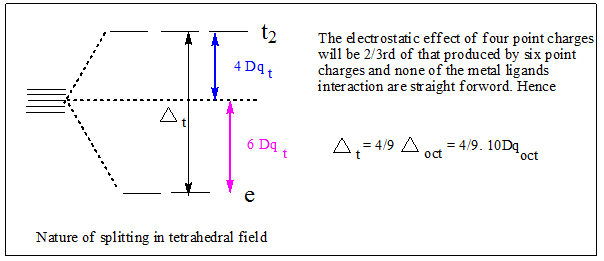
Q. Why most of the tetrahedral complexes are high spin in nature?
Answer: since tetrahedral complexes are marked by lower crystal field splitting, the energy separation is usually lower than the pairing energy.
Q. Most of tetrahedral complexes are high spin but [Cr{N(SiMe 3 ) 2 } 3 NO] is a low spin complex. Why?
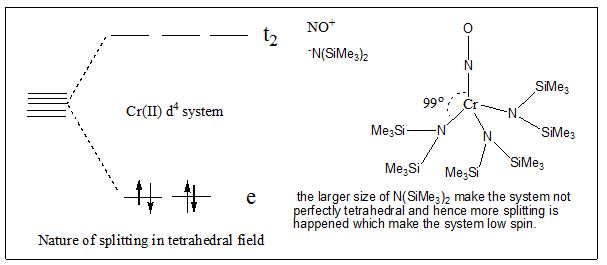
Or
Q. what's the magnetic behaviour of [Cr{N(SiMe3)2}3NO] or Tetrakis(1-norbornyl)cobalt(IV) i.e. diamagnetic or paramagnetic? Explain.