Crystal field stabilization energy:
The crystal field stabilization energy (CFSE) is the stability that results from placing a transition metal ion in the crystal field generated by a set of ligands. It arises due to the fact that when the d -orbitals are split in a ligand field (as described above), some of them become lower in energy than before with respect to a spherical field known as the bary-center in which all five d -orbitals are degenerate. For example, in an octahedral case, the t2g set becomes lower in energy than the orbitals in the bary-center. As a result of this, if there are any electrons occupying these orbitals, the metal ion is more stable in the ligand field relative to the bary-center by an amount known as the CFSE. Conversely, the eg orbitals (in the octahedral case) are higher in energy than in the bary-center, so putting electrons in these reduces the amount of CFSE.
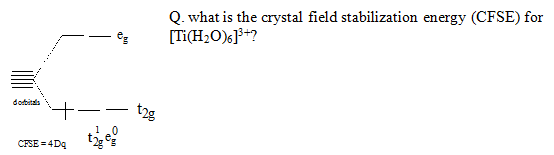
The only one d electron of Ti3+ will occupy the lowest available orbital & it is stabilized by the energy 4 Dq.
Q. what will be the electron arrangement for d4 and more than d4 conformation?
Answer: This splitting is affected by the following factors:
- the nature of the metal ion.
- the metal's oxidation state. A higher oxidation state leads to a larger splitting.
- the arrangement of the ligands around the metal ion.
- the nature of the ligands surrounding the metal ion. The stronger the effect of the ligands then the greater the difference between the high and low energy d groups.
There are two possibilities for these type of cases: e.g. for d4 case
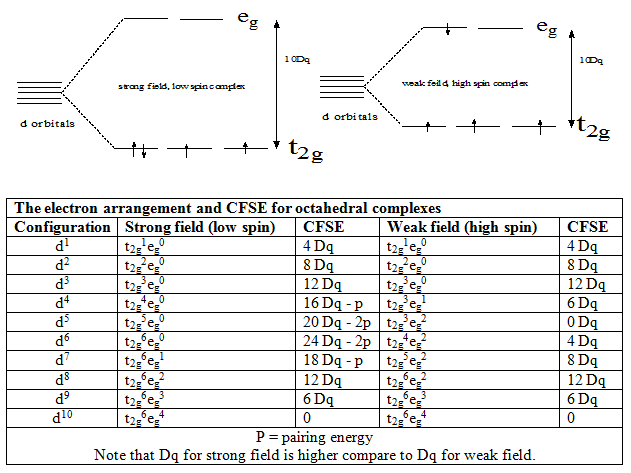
Effect of ligands on d orbital splitting ( spectrochemical series):
The ligands are sequencing according to their splitting ability, strong field ligand split the d orbitals more ( i . e . the separation between t 2g and e g is large) compare to weak field ligand and form low-spin complex where as weak field ligand form high spin complex.
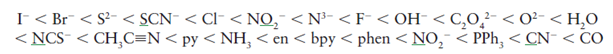
Q. What is the probable electron configuration of an octahedral complex [Co(CN)6]4− whose experimental magnetic moment is 4.0 BM?
A) t2g4 eg2 B) t2g6 eg1 C) t2g5 eg2 D) t2g6 eg2
Answer: ( C )
Referance/Hints: Co(II) is a d7 species and CN− is a strong field ligand. Hence a high spin configuration t2g5 eg2 is formed and the three unpaired electron give up the spin only magnetic moment 3.9 BM. (some amount of orbital contribution make it 4.0 BM)