7) Oxidation state of the central ion: Roman numerical (such as II, III, IV) at the end of metal part without any spacing.
![]()
8) Bridging group: µ- is written as a prefix of the ligand name.
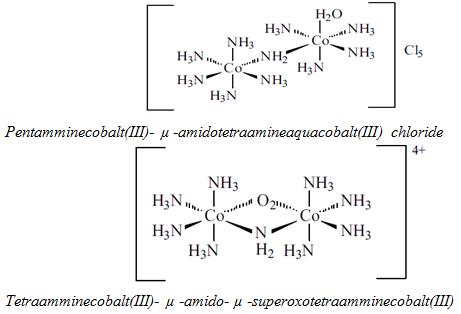
9) Point of attachment: if there is more than one atom for attachment with metal then the symbol of atom is written after the name of ligand.
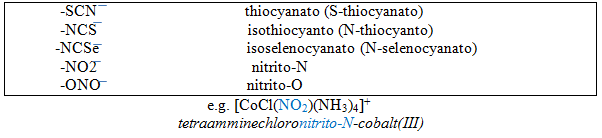
10) Isomer:

Q. You can have a compound where both the cation and the anion are complex ions. Notice how the name of the metal differs even though they are the same metal ions. What's the name of that type of compound e.g. [Ag(NH3)2][Ag(CN)2] ?
Answer: diamminesilver(I) dicyanoargentate(I)
Q. Write the name of the following complexes:
- [CoCl3(NH3)3]
- [Co(ONO)3(NH3)3]
- [Fe(ox)2(H2O)2]−
- Ag2[HgI4]