The value of ∆H has been found negative & comparable for three reactions. If we consider number of reactants and products for each of the three reaction, it is clear that Δ∆S for reaction (I) is zero in going from reactant to product side and the order of entropy change in going from reactant to product side is ΔS reaction(I)< ΔS reaction(II)< ΔS reaction(III) .
Note that ∆ ΔS is the measure of disorder of a system.
The above discussion shows that ΔG has a negative value for all three reactions and the magnitude of –ve value increases as the order ΔGreaction(I)< ΔGreaction(II)< ΔGreaction(III) .
So the stability order follow as the sequence [Cd(trien)]2+ > [Cd(en)2 ]2+>[Cd(CH3NH2)4]2+ which indicate the higher stability of the chelate complexes.
Nomenclature of coordination compounds:
1) Orders of naming ions in coordination complexes :
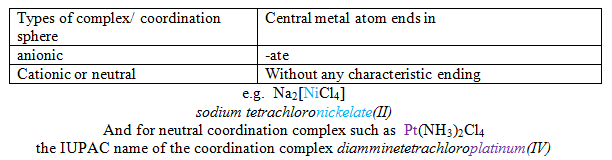
The names of neutral coordination complexes are given without spaces and for ionic coordination complexes the cation is named first & then the anion separated by a space.
![]()
Here the coordination complex is neutral, so no spaces are necessary. The word ‘ mer' is used for expressing the geometry.
![]()
In the above examples, the cations sodium is named first and then separated by a space from the names of the anions
2) Naming the coordination sphere: ligands are named first & then the metal ion.
![]()
3) Names of the ligands: Negative ligands end in –o & the positive ligands end in –ium. The neutral ligands are named as such.
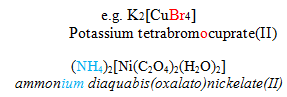
4) Order of naming the ligands: alphabetical order irrespective of their charge. ( IUPAC convention)
![]()
5) Numerical prefix to indicate the number of ligands:

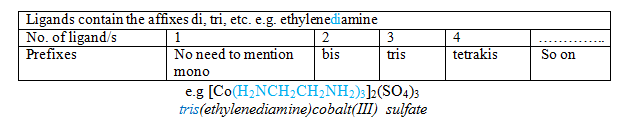
Remember that you never have to indicate the number of cations and anions in the name of an ionic compound .
6) Ending of names: