(iii) Electronpairs in filled shell repel stronger that electronpairs in incomplete shell.
When the electron pair containing atom contains low-energy lying suitable vacant orbitals the electron pair of electrons can undergoes diffusion to the vacant orbitals. Hence, the lp - bp repulsion diminishes dramatically and bp - bp repulsion dominates. In this case, the bond angles around the central metal increase.
Example:

- On going from O to Te the size of the central atom increases. Therefore, diffusion is more predominating which further decreases lp - bp repulsion.
(IV) Lone pair of electron can be transferred from a filled shell to a energetically suitable empty shell of the other bonded atom.
Due to transfer of lone pair of electron the bond accumulate some sort of double bond character. This enhances the bp - bp repulsion and consequently, causing a bigger bond angle.
Example:
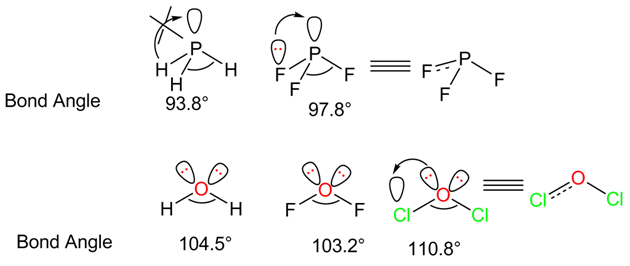
- P and Cl contain low-lying vacant d orbitals. Lone pair of electron from the filled orbitals of F or O can be transferred to the vacant orbital. This causes a partial - multiple bond that enhances the bp - bp repulsion. Hence, the bond angle increases.
(V) Multiple bond orbitals repel each other more strongly than single bond orbitals.
Examples:
O = PF3 (F-P-F bond angle = 103°), while in O = PBr3 (Br-P-Br bond angle = 108°)