The ionic bond:
As stated earlier, Ionic bonds are formed when electropositive elements interact with electronegative elements. The resulting compound is known as ionic compound.
Some features of ionic compounds:
(i) Ionic compounds tend to have low ionic conductivity in solid state but highly conducting in molten state. This is because in the solid state ions are held tightly by the surrounding counter ions (lattice effect) so, they are not free to move in the presence of any external electric field, whereas, in molten state it becomes easier for the movement of ions.
(ii) Ionic compounds are high melting, hard and brittle substances.
(iii) Ionic compounds are often soluble in polar solvent with high dielectric constants. The energy of interaction of two charge particles is given by;
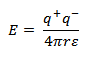
Where q + and q − are the charges, r is the separation distance, ε is the dielectric constant of the medium.
Radius ratio rules:
In ionic compounds, ions are held together by electrostatic attraction between two oppositely charged ions. The structures of ionic compounds depend on the relative size of positive and negative ions. The accommodation of the number of ions around a particular ion, i.e. coordination number, can be found out by geometric calculation.
Coordination number 3 (trigonal planar):
Let say, AB is an ionic compound having coordination number 3. All the three B are in contact with A but not to each other (Figure [1]). A limiting case arises when all the three are also come in contact with each other. Assume that the radius of cation is r + and anion is r -
Therefore, cos 30 ° = DC/DE
or, DE = DC / cos 30 °
or, r + + r − = r − / 0.866 [ ∴ cos 30 ° = 0.866]
or, r + + r − = 1.155 × r −
or, r + / r − = 1.115-1.000 = 0.155
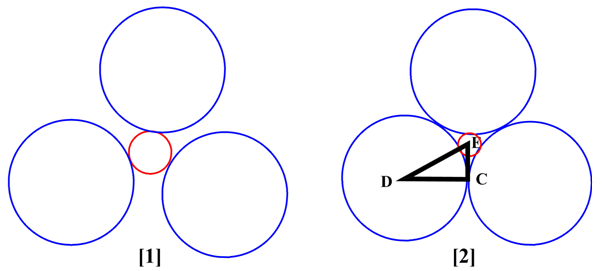
Figure 2.2. Non-limiting and limiting radius ratio for coordination number 3. Red circle represents cation and blue circles represent anion, respectively.
Simple geometry provides the limiting radius ration for coordination number 3 is 0.155.
When the radius ratio is smaller compared to 0.155, the positive ions will not be in contact with the negative ions, and therefore, a whole will be created and the resulting structure will be unstable. If the radius ration value is more than that of 0.155, it is possible to have contact between positively charged cation and a negatively charged anion. In this case the complex will be stable.
Coordination number 4 (tetrahedral):
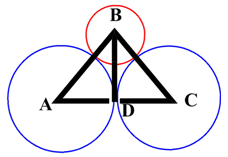
Figure 2.3. Cross section of a tetrahedral arrangement.