4.7.3. Mylotarg
4.7.3.1. Introduction
Because of lack of tumor-cell specificity and extreme cytotoxicity, the direct application of the enediyne antibiotics as antitumor drugs is generally limited. It is that need which drew attention of scientists to generate modified enediyne compounds with improved specificity and pharmacological properties.
Mylotarg (CMA-676, gemtuzumab ozogamicin), the combination of Calicheamicin (CAL) with an antibody that binds to CD33 antigen, was approved by the FDA in 2000 to treart acute myeloid leukemia (AML), a bone marrow cancer. It is the first in a new class of antibody-targeted chemotherapy for the treatment of patients 60 years and older in first relapse with CD33-positive acute myeloid leukemia (AML) ( Figure 5 ).
Mylotarg® composed of a recombinant humanized IgG4, kappa antibody conjugated with a cytotoxic enediyne antitumor antibiotic, calicheamicin. The antibody portion of Mylotarg binds specifically to the CD33 antigen. CD33 antigen is a sialic acid-dependent adhesion protein that is found on the surface of leukemic blasts and immature normal cells of myelomonocytic lineage. But it is not present in normal hematopoietic stem cells.
The anti-CD33 hP67.6 antibody is produced by mammalian cell suspension culture using a myeloma NS0 cell line. This is then purified to remove or inactivate viruses. Three separate and independent steps are involved in retrovirus inactivation and removal (in the purification) process and thus to get purified hP67.6 antibody. These steps include- (a) low pH treatment, (b) DEAE-Sepharose chromatography, and (c) viral filtration.
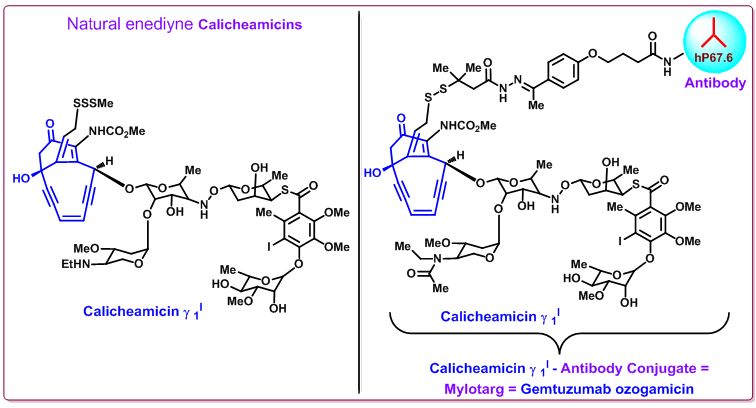
Figure 5. Structures of Calicheamicin and its antibody conjugate (Mylotarg).
Mylotarg contains amino acid sequences of which approximately 98.3% are of human origin. The constant region and framework regions contain human sequences while the complementarity-determining regions are derived from a murine antibody (p67.6) that binds CD33. This antibody is linked to N -acetyl-γ-calicheamicin via a bifunctional linker. Gemtuzumab ozogamicin has approximately 50% of the antibody loaded with 4-6 moles calicheamicin per mole of antibody. The remaining 50% of the antibody is not linked to the calicheamicin derivative. Gemtuzumab ozogamicin has a molecular weight of 151 to 153 kDa.
Mylotarg is a sterile, white, preservative-free lyophilized powder containing 5 mg of drug conjugate. The drug is light sensitive and must be protected from direct and indirect sunlight and unshielded fluorescent light during the preparation and administration of the infusion. The inactive ingredients are: dextran 40; sucrose; sodium chloride; monobasic and dibasic sodium phosphate.
Gemtuzumab interferes with the growth of cancer cells and slows their growth and spread in the body Gemtuzumab is usually given to people who are at least 60 years old and have a relapse of their disease and who cannot receive other cancer medications.
4.7.3.2. Mechanism of Action of Mylotarg
Gemtuzumab ozogamicin binds to the CD33 antigen. This antigen is expressed on the surface of leukemic blasts in more than 80% of patients with acute myeloid leukemia (AML). CD33 is also expressed on normal and leukemic myeloid colony-forming cells, including leukemic clonogenic precursors. But it is not expressed on pluripotent hematopoietic stem cells or on nonhematopoietic cells.
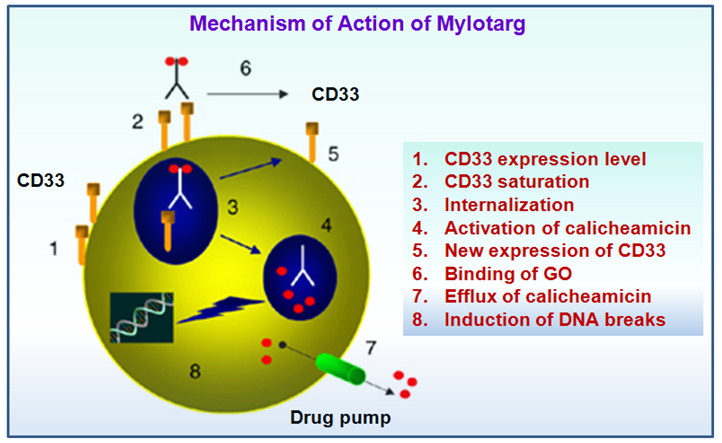
Figure 6. Schematics of mechanism of action of mylotarg
Mylotarg (Gemtuzumab ozogamicin) is first directed against the CD33 antigen expressed by hematopoietic cells. Then the anti-CD33 antibody portion of Mylotarg binds with the CD33 antigen. After infusion, near complete saturation of CD33 antigenic sites by Mylotarg was reached for AML blasts, monocytes, and granulocytes, whereas Mylotarg did not bind to lymphocytes.This binding event results in the formation of a complex that is internalized. Upon internalization, the calicheamicin derivative of Mylotarg is released inside the lysosomes of the myeloid cell. As soon as the enediyne anticancer antibiotic calicheamicin is released, it migrates to the cell nucleus and binds to the minor groove of leukemic DNA. Then the calicheamicin cleaves DNA double strand that ultimately lead to cell death.