4.7. The Approved Enediynes for Use as Anticancer Drugs
4.7.1. Enediyne Derivative and Conjugate in Clinical Use
Table 3: Enediyne derivatives/conjugates in Clinical Use

4.7.2. Neocarzinostatin (NCS)
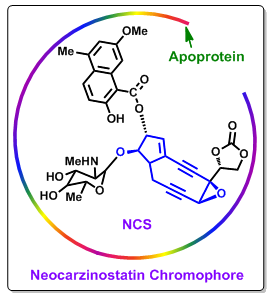
NCS is the first enediyne antitumor antibiotic that is in clinical use for the treatments of leukemia, gastric carcinoma and pancreatic adenocarcinoma (Figure 3). In clinical studies NCS is shown to be active against acute leukemia. Studies revealed that NCS give remedy of 9 out of 51 patients completely and of 9 a partial remedy. NCS also is found to effective against hepatoma and hematologic malignancies.
Both continuous and intermittent intravenous infusion was the process of NCS administration to patients with a variety of malignant diseases. Leukemic patients on intermittent therapy showed greater changes in bone marrow cellularity than those treated by continuous infusion.
Figure 3. Chemical structure of neocarzinostatin. |
Anorexia, nausea, and vomiting were the most frequent side effects of NCS administration. In phase I and phase II evaluations, dose limiting toxicity was late myelosupppression. Myelosuppression was more pronounced in patients who had received previous chemotherapy. Gastrointestinal side effects were mild. Three patients had a severe acute reaction resembling anaphylaxis. Allergic reactions were more frequent with intermittent than with continuous infusions. Therefore, the clinical trials of NCS were obstructed by anaphylactic responses.
4.7.2.1. Polymer-Neocarzinostatin Conjugate (SMA-NCS)
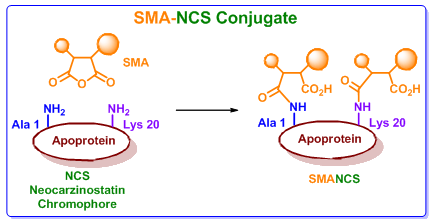
NCS is actually a prodrug that requires sulfhydryl activation for the activity (Figure 3), which results in lower selectivity and cytotoxic activity. However, initial clinical trials were hindered by anaphylactic responses due to its non-covalently bound protein component. It was observed that if NCS could be made immunologically inert then this allergic action can be removed. This was exactly done by coupling to a maleic acid-based polymer (SMANCS, Figure 4). The SMANCS is thus clinically used primarily in the treatment of hepatocellular carcinoma. Hepatocellular carcinoma (HCC, also called malignant hepatoma) is the most common type of liver cancer. Most cases of HCC are secondary to either a viral hepatitis infection (hepatitis B or C) or cirrhosis (alcoholism being the most common cause of hepatic cirrhosis).
A principal advantage in the use of SMANCS is the tumor targeting mechanism through an enhanced permeability and retention effect. Use of SMANCS has resulted in potential reduction or elimination of toxicity as is generally accounted in bone marrow toxicity associated with the use of NCS.
Figure 4. The SMA-NCS conjugate. |
SMANCS was used for the treatment of hepatoma, gastric carcinoma and lung cancer. This was approved in Japan . Since then, hepatic arterial infusion of a SMANCS/Lipiodol emulsion has been used as one of the practical treatments for advanced or recurrent hepatocellular carcinoma .
Phase I Clinical Evaluation: In Phase I, SMANCS was found to decrease the concentrations of α - fetoprotein in 86% of patients and decrease the tumour size in 95% patients.
Phase II Clinical Evaluation: Phase II clinical study with primary hepatoma also showed a
relatively high response rate of 36~40%.
The most successful use of SMANCS was seen when it was administered to an individual patient. In this treatment the SMANCS was administered as dose per tumour size and follow-up treatments were given on a need basis.
Arterial infusion therapy with SMANCS/Lipiodol was effective for large renal cell carcinoma without metastases in conjunction with surgery. In surgical patients without metastases administration of SMANCS/Lipiodol infusion has lead to the 5- and 10-year survival rates of 83.0% and 75.2%, respectively.