Total Synthesis
- • Meerwein-Ponndorf-Verley reduction of 5 could convert the keto group into hydroxyl that can displace on the carbonyl of the six membered lactone ring, giving a five membered lactone, and the hydroxyl group so released can open the epoxide ring to afford 6 (Scheme 3).
- • Esterification of the carboxyl group using diazomethane,acetylation of the alcohol group using Ac2O and dihydroxylation of the double bond can give 12 (xi) that could undergo oxidative cleavage followed by esterification of the new carboxyl group with diazomethane to give 13 (xii).
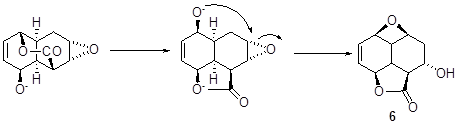
Scheme 3
• Dehydration of 6 can give α,β -unsaturated carbonyl compound 7 that could undergo conjugate addition at the less hindered α -side with methoxide to give 8 (vi and vii).
• NBS in acid could approach α -side of 8 to give a brominium ion that could be opened by water to give the biaxial bromo-alcohol 9 (viii) that could undergo mild oxidation to afford 10 (ix).
• Zn in AcOH can bring the reductive opening of both the lactone and the strained ether of 10 to give 11 (x) (Scheme 4).
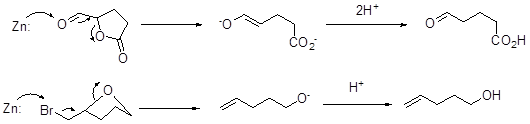
Scheme 4
• Schiff base formation of 13 with 6-methoxytryptamine can give 14 that could be converted into 15 by NaBH4 reduction of the imine double bond (xiii and xiv). Treatment of 15 with POCl3 can bring ring closure as in the Bischler-Napieralski synthesis of isoquinoline, providing an imminium salt 18 via 16 and 17 (xv), which could be reduced using NaBH4 to give 19 (xvi).
• Base hydrolysis of 19 can give 20 having free OH and COOH groups that could be joined to give a lactone 21 using DCC (xvii and xviii). Epimerization of the less stable 21 using t-butyric acid can give the required more stable 22 that could be converted into (±)-reserpine by opening of the lactone with MeOH followed by acylation using 3,4,5-trimethoxybenzoyl chloride. The (±)-reserpine could be resolved using CSA in a 3:1 mixture of MeOH and CHCl3 .