12.1.1 Retrosynthetic Analysis
The strategy was based on building five contiguous stereocentres into a decalin derivative that could be opened to a monocyclic compound to form ring E (Scheme 1).
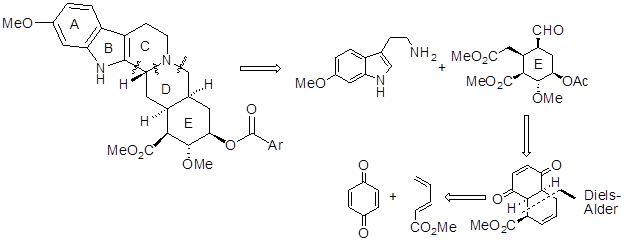
Scheme 1. Retrosynthetic Analysis
12.1.2 Total Synthesis
- • The Diels-Alder reaction can lead to the ring junction having cis stereochemistry and the carboxyl group lie on the same side as the rings with respect to the ring junction (i) (Scheme 2). This step fixes the stereochemistry at C 15, C 16 and C20 of reserpine.
• NaBH4 reduction of the less hindered of the two carbonyl groups of 2 can provide 3 (ii). The epoxidation of the isolated double bond with mCPBA at the less hindered side can afford 4 (iii) that could undergo dehydration to give the lactone 5 (iv).