3.9.2. Trypsin, Chymotrypsin, and Elastase: The Digestive Serine Proteases
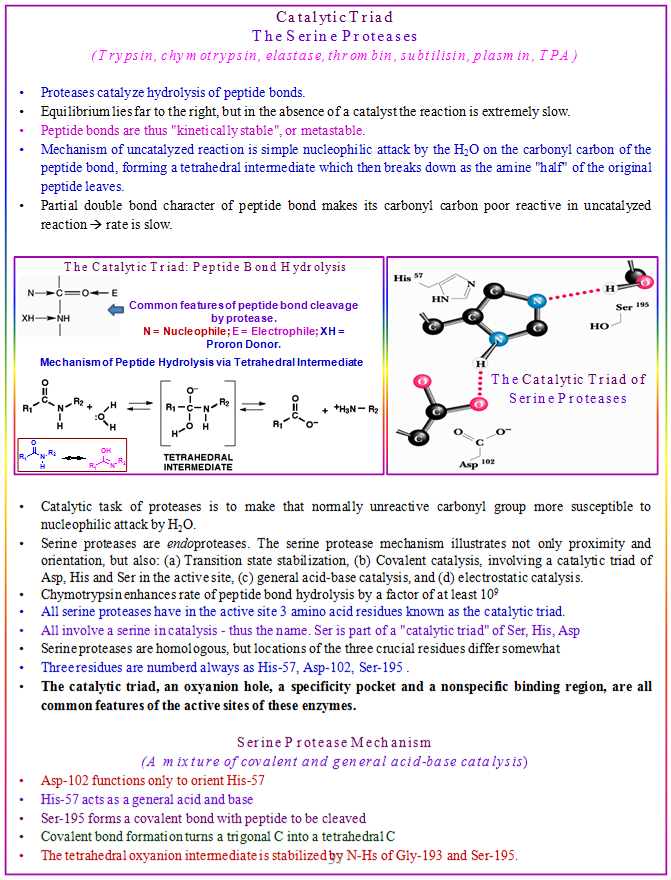
Trypsin, chymotrypsin, and elastase all carry out the same reaction — the cleavage of a peptide chain — and although their structures and mechanisms are quite similar, they display very different specificities. Trypsin cleaves peptides on the carbonyl side of the basic amino acids, arginine or lysine. Chymotrypsin prefers to cleave on the carbonyl side of aromatic residues, such as phenylalanine and tyrosine. Elastase is not as specific as the other two; it mainly cleaves peptides on the carbonyl side of small, neutral residues. These three enzymes all have similar sequences and three-dimensional structures. In Chymotrypsin, Serine195 binds to the substrate polypeptide to the side of a phenylalanine, tryptophan, or tyrosine residue closer to the C-terminus, holding it in place. Aspartate (D102) and Histidine (H57) then hydrolyze the bond.