3.9. The Catalytic Triad
Proteases, commonly called peptidases, represent approximately 2% of the total number of proteins present in all types of organisms. There are about 500 human genes that encode peptidases and their homolog. Many of these enzymes are of medical importance, and are potential drug targets that originate from the human genome or from the genome of the disease-causing organism.
Classification of Proteases: The homologous peptidases are divided into clans and families. Families are grouped in a clan according to tertiary structure comparisons. Clans arise from a common ancestor. Four distinct mechanisms are known that linked to the serine, cysteine, aspartic and metallopeptidases, respectively. The most conspicuous difference in the mechanisms is the presence or absence of a covalent acyl-enzyme intermediate on the reaction pathway.
Catalysis by Proteases: The catalyses of serine and cysteine peptidases involve the covalent intermediate (ester and thiolester, respectively). During hydrolysis carried out by aspartic and the metallopeptidase catalyses, the substrate is attacked directly by a water molecule rather than by a serine or cysteine residue. In spite of the differences, the basic catalytic features of the different clans are common. Hydrolysis of the peptide bond is an addition-elimination reaction involving a tetrahedral intermediate. All three heavy atoms of the peptide bond are directly implicated in the catalytic reaction by interacting with appropriate enzymatic groups.
3.9.1. The Catalytic Mechanism: The Catalytic Triad:
A catalytic triad refers to the three amino acid residues found inside the active site of certain protease enzymes: serine (S), aspartate (D), and histidine (H). They work together to break peptide bonds on polypeptides. In general terms, catalytic triad can refer to any set of three residues that function together and are directly involved in catalysis. Because enzymes fold into complex three-dimensional shapes, the residues of a catalytic triad are brought close together in the tertiary structure.
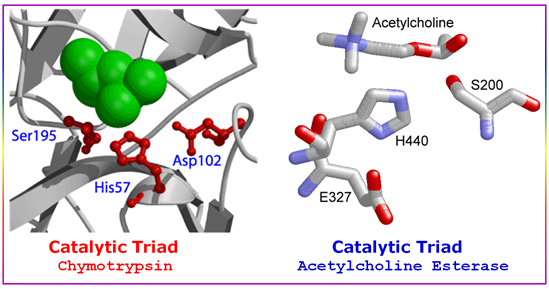
Figure 3.5: The Chymotrypsin and Acetylcholine Esterase Catalytic Triads.
Serine proteases are a class of proteolytic enzymes whose catalytic mechanism is based on an active-site serine residue. Serine proteases are one of the best-characterized families of enzymes. This family includes trypsin, chymotrypsin, elastase, (Digestive enzyme) thrombin (crucial enzyme in the blood-clotting cascade), subtilisin (bacterial protease), plasmin(breaks down the fibrin polymers of blood clots), tissue plasminogen activator (cleaves the proenzyme plasminogen, yielding plasmin), and other related enzymes. Finally, although not itself a protease, acetylcholinesterase is a serine esterase and is related mechanistically to the serine proteases. It degrades the neurotransmitter acetylcholine in the synaptic cleft between neurons.