1.1.5.3.2.3.2. Selective Oxygenation of Saturated C-H Bonds by a Dimanganese Catalyst:
Although enzymes often incorporate molecular recognition elements to orient substrates selectively, such strategies are rarely achieved by synthetic catalysts. Robert H. Crabtree, Gary W. Brudvig, et. al. combined molecular recognition through hydrogen bonding with C-H activation to obtain high-turnover catalytic regioselective functionalization of sp3 C-H bonds remote from the –COOH recognition group. The catalyst contains a Mn(m-O)2Mn reactive center and a ligand based on Kemp’s triacid that directs a –COOH group to anchor the carboxylic acid group of the substrate and thus modify the usual selectivity for oxidation. Control experiments supported the role of hydrogen bonding in orienting the substrate to achieve high selectivity.
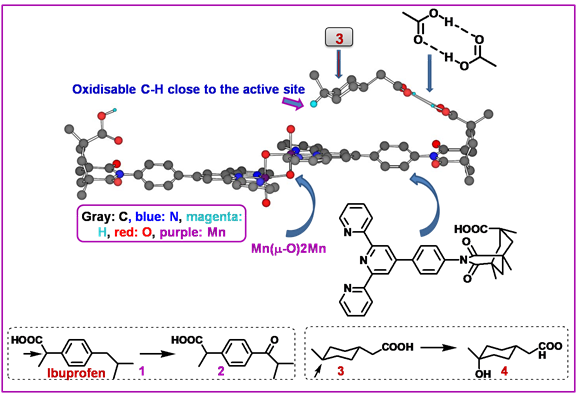
Figure 1.9: The intermediate, resulting from H-atom abstraction from C6 of 3 in a distorted chair conformation with its Manganese complex (Gray: C, blue: N, magenta: H, red: O, purple: Mn)
1.1.5.3.2.3.3. Enzyme Models for Transamination
1.1.5.3.2.3.3.A. Introduction: Transamination
- Interchange of the functional groups between a α-keto acid and one amino acid.
- ENZYMES THAT CATALYZE THESE REACTIONS:
- Aminotransferases or transaminases
- REQUIRED COFACTOR
- Pyridoxal Phosphate: Pyridoxal Phosphate is the active form of Vitamin B-6. This vitamin has three active forms: pyridoxal, pyridoxine (or piridoxol) and pyridoxamine. Sometimes pyridoxine is used as synonym of Vitamin B6.
1.1.5.3.2.3.3.B. The Mechanism of Transamination
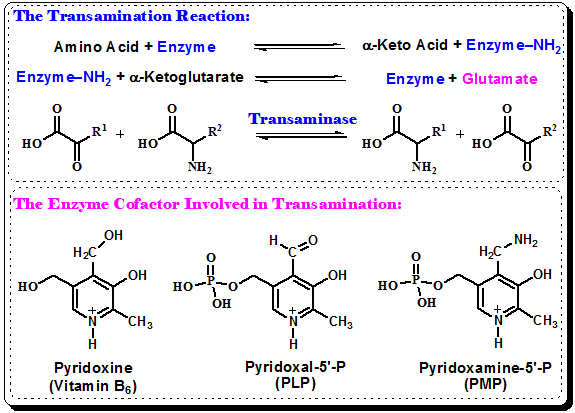
Figure 1.10: Mechanism of Transamination reaction.