1.1.5.3.2.2. Why Nature had selected the normal DNA structure?
Background from Biology: A study of cleavage of RNA by enzyme models, showed that the cleavage was accompanied by simultaneous isomerization of the natural 3',5'-linked RNA to its 2',5'-linked isomer. It is also established that ribonucleosides are converted to deoxyribonucleosides by a biochemical process that removes the 2'-hydroxyl group, and that could have removed the 3'-hydroxyl group instead with a different enzymatic preference. These observations raised the question of whether there is some intrinsic chemical preference for the natural structure of DNA, with its phosphate links joining the 3' and 5' positions of adjacent bases. Is the current preference simply an accident of evolution, or is there an intrinsic disadvantage to the use of a 3'-deoxy 2',5'-linked analog of natural DNA?
Application of Chemical synthesis to Answer: Professor Breslow and his group in 1990’s have prepared DNA isomers in which they have used 3'-deoxynucleosides, and which have the unnatural 2',5'-links, called iso-DNA. However, they observed that iso-DNA makes a much weaker double helix with its conjugate, or with the conjugate based on normal DNA, because the helix has more hydrophobic surface exposed to solvent. Thus, it is not a suitable substitute for normal DNA as a genetic material, and organisms that may have tried it would not be competitive. However, iso-DNA does make a strong heteroduplex with normal RNA, reflecting the conformation of the ribose ring relative to that in deoxyribose.
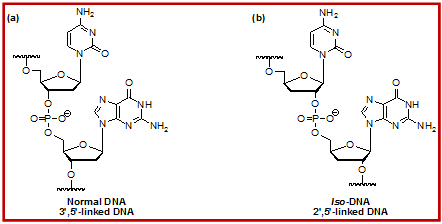
Figure 1.5: Chemical Structures of (a) natural 3', 5'-linked DNA and (b) unnatural 2', 5'-linked iso-DNA.
1.1.5.3.2.3. Lesson from Enzyme biology
Many inspiring principles were received from enzyme biology. As for an example geometry can dominate chemical reactivity. A good example is the conversion of lanosterol to cholesterol, in which three inactivated methyl groups are oxidatively degraded by enzymes of the class cytochrome P-450 while the much more reactive double bonds of lanosterol are left untouched until later (Fig. 6).
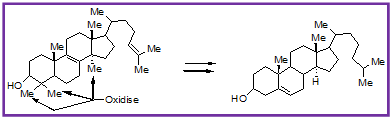
Figure 1.6: Conversion of lanosterol to cholesterol, showing oxidative degradation of three inactivated methyl groups by enzyme, cytochrome P-450.