Example: Potential Determining Ions
Till now we have looked at indifferent ions which do not change potential on surface. But when the surface consists of mobile ions (like H+ or  -ions) which can go from surface to solution, the surface acquires excess charge due to the movement of mobile ions into the solution. -ions) which can go from surface to solution, the surface acquires excess charge due to the movement of mobile ions into the solution.
For example, let's consider a surface consisting of AH type of molecules in an electrolyte. Now the AH molecule can dissociate into A- and H+ ions.
Further, if this AH molecule is part of another large molecule embedded in the underlying surface (like–COOH group at the end of a protein molecule), the H+ ions could move into the solution, leaving the surface with an excess negative charge at the 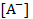 ions sites. ions sites.
If K is the equilibrium constant then,
where,
[AH] is the concentration of those sites which have not dissociated,
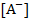 is the concentration of ions on the surface and, is the concentration of ions on the surface and,
[H+] is the concentration of hydride ions in the solution near the surface.
If [n0] is the total number of sites/dissociable molecules at the surface then,
Total no. of sites = no. of non-dissociated sites + no. of ions on the surface
Combining equations (10.7) and (10.8), to eliminate 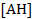 we have we have
Since the surface consists of single valence 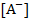 ions, the charge density could be written as: ions, the charge density could be written as:
|