| |
Now, the surface potential (or more precisely the zeta potential, 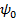 ) varies inversely with the valence of ion due to the irreversibly adsorbed counter-ions at the surface (also known as Stern layer). ) varies inversely with the valence of ion due to the irreversibly adsorbed counter-ions at the surface (also known as Stern layer).
Note that Boltzmann’s distribution for potential holds for free charge in the solution (diffused region). In case of adsorption of ions on the surface, we would have to define a new value of ψd outside the surface of the bound charges as shown in the figure.
Thus, again
which implies that Schultz-Hardy rule is valid for small potentials as well.
Let’s look at some experimental values in this regard:
Table 10.1: Some typical values for CCC for common metal ions
Valence of ion |
Example |
CCC (mmol/L) |
1 |
Li, Na, K |
50 |
2 |
Mg, Zn |
0.7 |
3 |
Al, Ce |
0.09 |
|