This charge density depends upon the association/dissociation equilibrium of the [H+] ions, which in turn depends upon their concentration near the surface. Higher [H+] near the surface indicates larger number of associated ions.
If we have HCl in bulk then there will be a bulk concentration of [H+]
Boltzmann distribution of these ions in solution is given by:
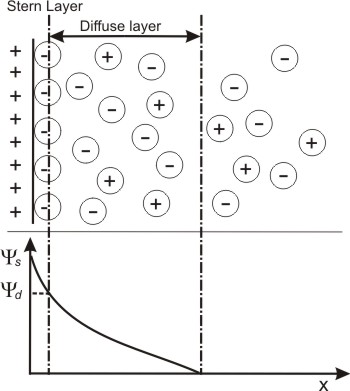
Fig. 10.5: Change in effective surface potential due to adsorption of ions on the surface
(Valence in this case is 1)
At the surface, 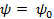 , Therefore , Therefore
Hydrogen ion is the counter ion in this case.
We can change [H+]SOLN to change [A-]
Now the question is that how do we determine ψo.
|