40.3 NYLON
Polymer Chemistry : Nylon 66 (or 6,6 - Nylon) is a condensation polymer formed by the condensation polymerization of Adipic acid (a dibasic acid) and hexamethylene diamine (a diamine). In the nomenclature, the first number refers to the number of carbon atoms in the diamine and the second number designates the number of carbon atoms in the dibasic acid.
Nylon – 6 is a polymer of caprolactum . The number refers to the total carbon atoms in the ring, which can vary from 5 to 12.
Both Nylon – 6 and Nylon – 66 have similar properties and can be used as thin films and fibres; and for extrusion and injection molding in plastics. The major difference in the physical properties of these two nylons is that the melting point of Nylon – 66 is 40 – 45°C higher than Nylon – 6. Moreover, Nylon – 66 is harder, rigid and its abrasive – resistance is lower than that of Nylon – 6.
Technology (Figure 40.3):
In the production of Nylon – 66, first, the amine and acid is mixed in an aqueous solution , with the pH adjusted at 7.8 to form Nylon salt. The salt is then concentrated under vacuum. It is thereafter charged to an autoclave where the rest of the water is removed. The temperature is gradually increased to 280 o C in the autoclave to complete the polymerization and the water of condensation is removed.
The final product has a molecular weight of 12000 – 16000. It is extruded as ribbons onto chilling rolls. Then, it is sent to a chipper which produces small chips, which possesses the convenience of storage and rehandling. These chips are melted, metered through high pressure pumps. After filtration, these are passed via a melt spinnerette to produce nylon fibers. Upon cooling , these filaments harden and are wound on bobbins at a rate of 750 metres per minute or even higher. Inorder to give the fibre desirable textile properties, the threads are stretched to about 4 times its original length.
The production of Nylon – 6 is similar to that of Nylon – 66 , the only difference being the nature of polymerization. Nylon – 6 is manufactured by the step – wise condensation of caprolactum with no net water removal. However, to maintain the thermal equilibrium between monomer and polymer at the melting and spinning temperatures is the only difficult task. Monomer retained on the fibres accounts upto 10% , so the fibres are water – washed to remove the soluble caprolactum retained.
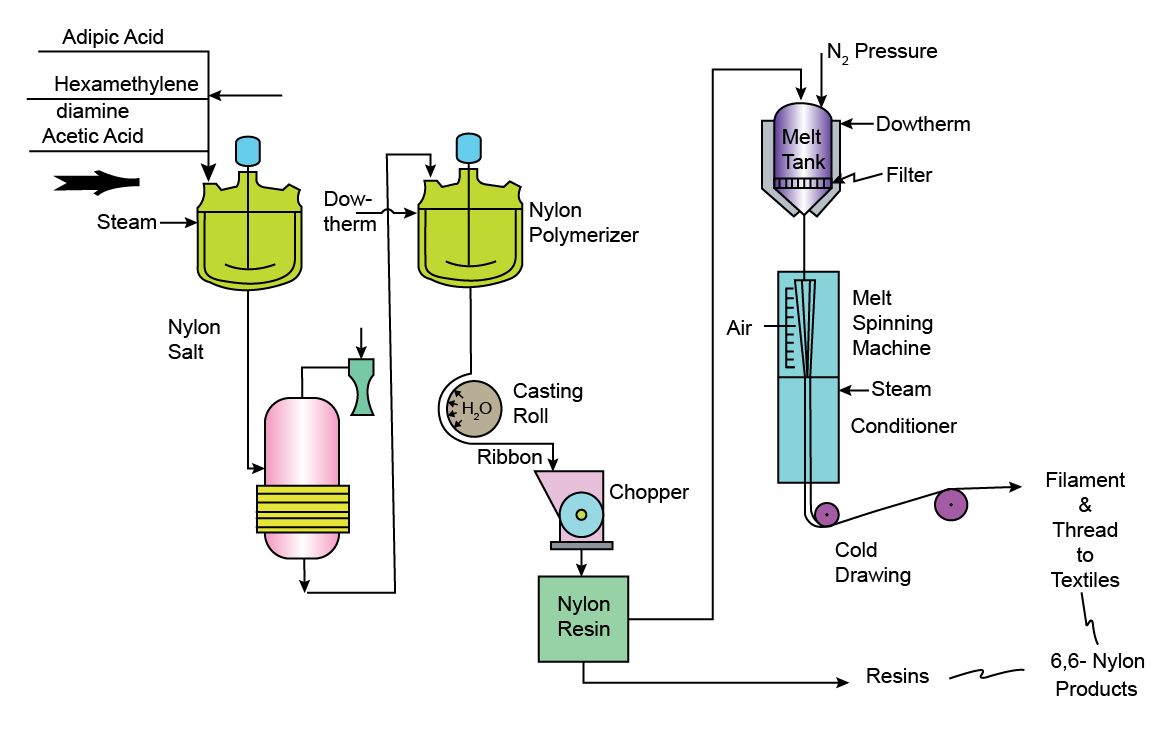
Figure 40.3 Manufacture of Nylon