31b.1 Hydrogenation of oil
Hydrogenation process is used to remove double bonds and to make fats and oil saturated. Hydrogenation also raises its melting point and improves its resistance to rancid oxidation. The most common end product of hydrogenation is Vanaspati ghee. Other products include vegetable ghee, hardened industrial oils and partially hydrogenated liquid oil.
Chemical Reactions: (* indicates activated catalytic state)
....................................Ni* catalyst
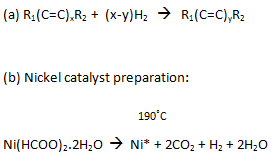
This produces a finely divided catalyst which is preferred for well-stirred hydrogenation reactors.
(c) Nickel catalyst preparation (reduced Ni on inert catalyst support)
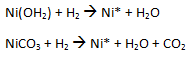
Nickel salts are precipitated on inert porous carrier such as kieselguhr or diatomaceous earth and reduced at high temperature in a hydrogen atmosphere.
(d) Nickel catalyst preparation
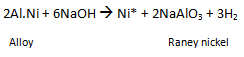
Produces spongy, high-surface area catalyst when the sodium aluminate is washed with water.