9.1 Introduction
- • In this lecture we present a brief overview of the alkylation process.
• In an alkylation process, olefins are reacted with isoparaffins to yield alkylate product.
• The basic purpose of alkylation is to enhance the octane number of the feed stock.
• For instance, octane number of butane alkylate is about 92 – 97. This is due to the formation of a hydrocarbon with side chain arrangement of carbon and hydrogen atoms.
9.2 Reaction Mechanism
- • There are three basic reaction steps to achieve alkylation
• Step 1 involving Carbonium ion formation: In this reaction, alkene reacts with a proton (acid catalyst) to produce a proton substituted olefin. The proton substituted olefin reacts with isoparaffin to generate a reactive carbonium ion and alkane.
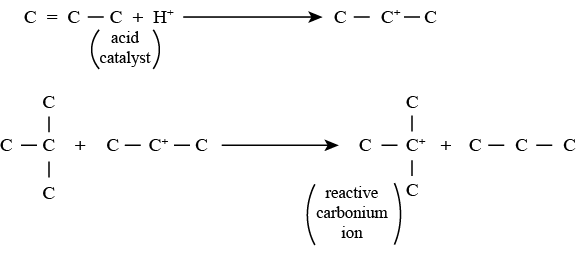
• Step 2 involving carbonium ion intermediate formation: In this reaction, the carbonium ion formed in step 1 reacts with the olefin to produce an intermediate carbonium ion.
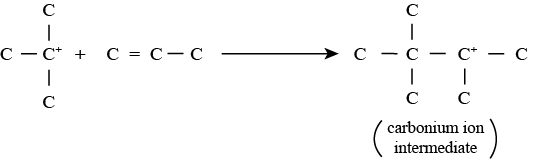
• Step 3 involving regeneration of carbonium ion: In this reaction, the intermediate carbonium ion reacts with the isoparaffin to produce alkylate product and carbonium ion. Thus carbonium ion is again regenerated to take part in step 2 reactions along with other additional unreacted olefin molecules.
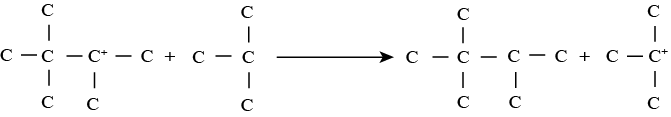
9.3 Reaction conditions
- • To avoid olefin polymerization, high isobutane to olefin ratios are used.
• Typical isobutene to olefin ratios are 5:1 to 15:1
• Acid catalysts are used. Primarily sulphuric acid (H2SO4) or HF are used.
• Depending on the acid catalysts choosed the process complexity varies. We present both process technologies to indicate the pertinent process complexity.
• Reaction operating temperature: 10 - 20°C using H2SO4 and 25 – 40°C using HF
• Reaction temperature: 4.4 bar for H2SO4 and 7.8 bar for HF
• When H2SO4 is used refrigeration is used.
• When HF is used, refrigeration is not used.
9.4 Sulfuric acid based alkylation process technology (Figure 9.1)
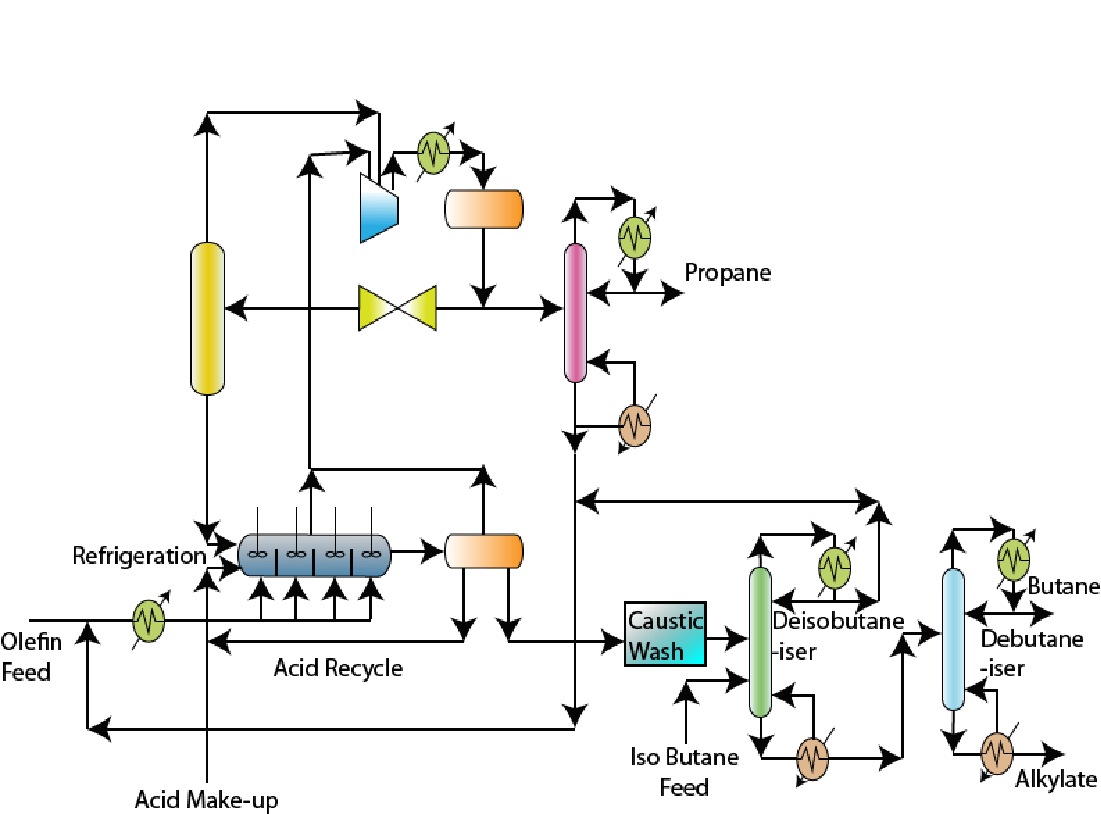 |
Figure 9.1 Sulphuric Acid Alkylation Unit
- • Caustic wash: The feed mixture (olefin + C4 compounds) are first subjected to caustic wash. During caustic wash, sulphur compounds are removed and spent caustic is recycled back to the caustic wash. Fresh caustic solution is added to take care of the loss.
• Refrigeration: The olefin feed enters a refrigeration unit to reduce the feedstock temperature.