7.4 Process parameters
- • Reactor pressure: 4 – 24 barg
• Reactor temperature: 500 – 525°C
• H2/Hydrocarbon molar ratio: 2 – 3
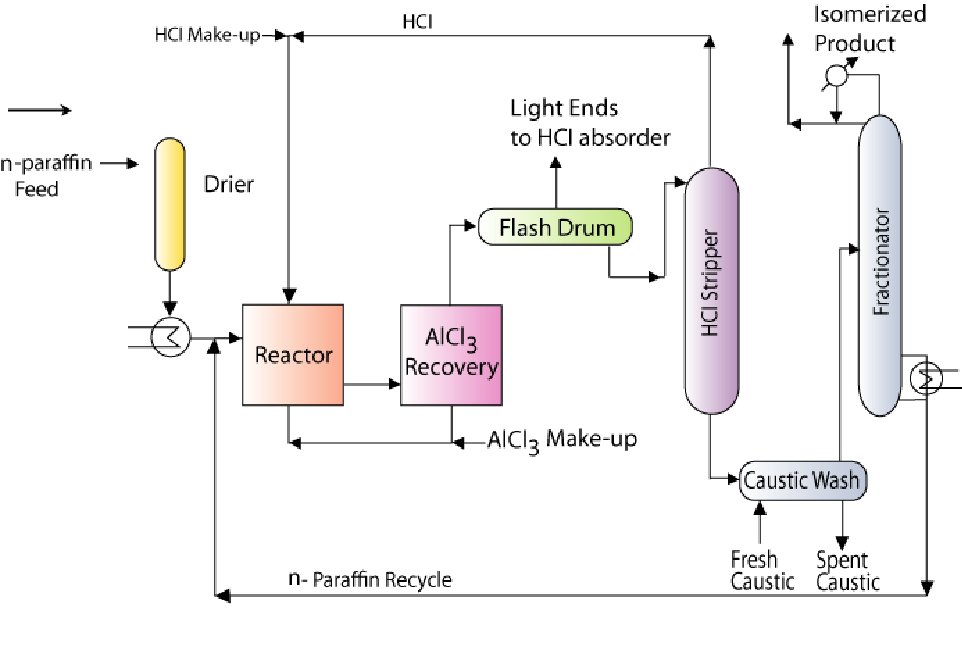
Figure 7.2 Flow sheet of Isomerization of n-paraffin
7.5 Introduction
- • The basic principle of Isomerization is to straight chain alkanes to side chain paraffins. This enhances the octane number substantially
• For instance, n-pentane has an octane number of 61.7 where as iso-pentane has an octane number of 92.3
• Usually light naphtha is used as a feed stock
7.6 Catalyst
- • Platinum base catalysts are used
• AlCll3 is used as a promoter for the catalyst
• During reaction, part of the AlCl3 gets converted to HCl
• Therefore, completely dry conditions shall be maintained to avoid catalyst deactivation and corrosion.
• Catalytic reaction takes place in the presence of hydrogen to suppress coke formation
7.7 Process technology (Figure 7.2)
- • Light naphtha and hydrogen (make up) are totally dried and sent to an isomerisation reactor after pre-heating the feed mixture in a heat exchanger
• Reaction operating conditions: 150 – 200°C and 17 – 28 barg
• Typical conversions: 75 – 80 % for pentanes.
• After reaction, AlCl3 is recovered from the product using condensation or distillation
• The basic principle for AlCl3 recovery is that at the reactor operating conditions, the AlCll3 is in volatile conditions and is soluble in hydrocarbons
• After AlCl3 is recovered from the product, it is sent back to the reactor along with the make- up AlCl3
• Eventually, the product enters a flash drum where bulkly light ends along with little quantities of HCl are separated from the liquid product.
• The light ends recovered from the flash drum are sent to a HCl absorber where HCl is absorbed into caustic solution to generate the light end gases. The light end gases can be further used for other processes in the refinery.
• The bottom product then enters a HCl stripper where most of the HCl is stripped from the isomerisation product rich stream. The HCl is recycled back to the reactor to ensure good catalyst activity. Make-up HCl is added to account for losses
• Subsequently, caustic wash is carried out to remove any trace quantities of HCl
• The isoermized product rich stream is then sent to a fractionators that separates the isomerized product from the unreacted feed.
• The unreacted feed from the fractionators is sent back to the reactor.