Reverse micelle method
Reverse micelles are formed when surfactant molecules, possessing polar head group and hydrophobic tail, are placed in non-polar solvent. In reverse micelle structure surfactant molecules aggregate into nano sized spherical structures having core of polar head groups and shell of hydrophobic tails. If small amount of polar solvent is present in the mixture, it would be enclosed in core of reverse micelles. If this polar solvent contains an oxide precursor then controlled hydrolysis of precursor will be achieved by mixing small amount of water and small particles of metal oxide will form at core. Preparation of nano Al2O3 by reverse micelle method is discussed below.
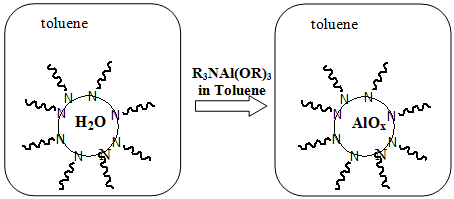
Fig 2. Schematics showing formation of nano-sized particles of aluminium hydroxide by reverse micelle method.
A solution of inverse micelles is first formed by adding a long chain alkylamine to a toluene solution. A small amount of water is trapped in the reverse micelle core. Mixing the reverse micelle solution with an aluminium alkoxy amine adduct results in hydrolysis of the aluminium alkoxide adduct and formation of nano-sized particles of aluminium hydroxide after drying.
Reverse micelle synthesis method is also reported for preparation of supported metal catalysts. Cheney et al. [5] reported synthesis of alumina supported Pt/Ni bimetallic catalysts by reverse micelle synthesis method. In this method two microemulsions were created by mixing 15% water, 10% surfactant and 75 % hydrocarbon( cyclohexane and propanol). Ni and Pt precursors were added to microemulsion -1 and hydrazine to the microemulsion -2. Each mixture was stirred separately for 1 h to allow micelles to equilibrate. In microemulsion-1 reverse micelles were formed having aqueous core and the Pt and Ni precursors were dissolved in aqueous polar core of the reverse micelles. Thereafter, the microemulsion –2 containing the reducing agent was added to the microemulsion -1 for insitu chemical reduction of the metals at reverse micelle core. The alumina support was added to the solution and titrated with acetone to disrupt the micelles and precipitate the nanoparticles onto the support. The supernatant was decanted and the catalyst powder was rinsed with acetone. Residual surfactant was removed by giving heat treatment in oxygen environment. By this method particles were obtained in the range of 1.4-2.8 nm.
Application of nanocatalysts
- Gold nanocatalysts
Supported gold nanoparticles have been reported to be very effective catalysts for oxidation of alcohols. Gold supported on graphite can oxidize glycerol to glycerate under relatively mild conditions. Under same conditions conventional supported Pt or Pd oxidation catalysts are much less selective.
- Sulfated zirconia nanocatalysts
These catalysts are highly effective in refining processes replacing liquid acid catalysts. These are used in acid promoted catalytic reactions such as alkylation,dehydration, isomerization etc.
- Dendrimer encapsulated nanoparticles (DENs)
Pd-DENs catalysts are studied as carbon-carbon coupling catalysts. In Heck coupling reaction, involving C-C coupling between aryl halides or vinyl halides and activated alkenes in the presence of a base, the Pd-DEN catalysts show higher activity and selectivity relative to other colloidal Pd catalysts.
- Tungsten oxide and titanium oxide nanocatalysts
Tungsten oxide and titanium oxide due to their visible-ultraviolet band gap have been extensively studied in electro-catalytic and photocatalytic applications.
- Transition metal or metal oxide nanoparticles
Transition metal or metal oxide nanoparticles have found important application in catalyzing growth of carbon nanotubes, carbon fibres, nanowires etc. Cobalt or cobalt oxide nanoparticles mixed with Ni is used in synthesis of single walled carbon nanotubes.
Text Reference
- B. Zoung, S. Han, R. Raja, G.A. Somorjai (Eds.), Nanotechnology in catalysis, Vol 3, Springer Science 2007
- Yuri Gogotsi (Ed.) Nanotubes and nanofibres, Taylor & Francis, 2006
Journal reference
- W.G. Menezes, V. Zielasek, K. Thiel, A. Hartwig, M. Bäumer, Journal of Catalysis 299 (2013) 222–231
- Y. Cheng , Y. Zheng , Y. Wang, F. Bao, Y. Qin, Journal of Solid State Chemistry 178 (2005) 2394–2397
- H. Xu, L. Zeng, S. Xing, Y. Xian, L. Jin, Electrochemistry Communications 10 (2008) 551–554
- Y. Men, M. Higuchi, K.Yamamoto, Science and Technology of Advanced Materials 7 (2006) 139-144
- B . A. Cheney, J. A. Lauterbach, J.G. Chen, Applied Catalysis A: General 394 (2011) 41–47