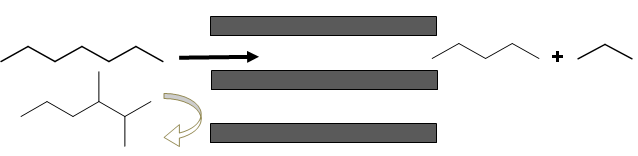
- Reactant selectivity: Selective admission of reactants to zeolite pores due to pore size restrictions is known as reactant selectivity. For example in case of cracking reactions, n-heptane undergoes preferential cracking (relative rate 1) over dimethylhexane (relative rate 0.09). The dimethylhexane, due to presence of branched carbons, is unable to enter the zeolite pores.
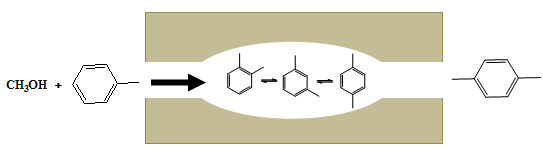
- Product selectivity: When there is possibility of formation of multiple products, selective formation of product can occur due to restriction on size or diffusion rates of the larger molecule. For examples when alkylation of methylbenzene is carried out over pentasil zeolites all isomers p-xylne, m-xylene, and o-xylene are probable products. However, due to pore diameter restrictions in pentasil zeolites there is preferential production of p-xylene over ortho and meta forms as shown in the figure below.
- Molecular traffic control: This concept involves preferential diffusion of reactants through one channel and diffusion of products out of another interconnecting channel of a zeolite. Counter diffusion is minimized and product selectivity is maximized by this process.