Zeolites
Composition and structures
Zeolites are crystalline aluminosilicates with pores of molecular dimensions . The general formula for a zeolite is Mx/n [(AlO2)x (SiO2)y].mH2O. M is the metal or hydrogen cation of valency ‘n' occupying the exchangeable cationic sites on zeolite framework. AlO2 and SiO2 are fundamental units sharing oxygen ions to form tetrahedral AlO4 and SiO4 building blocks for zeolite unit cell. Since silicon ion has +4 and Aluminium has +3 charges there is an overall negative charge on the aluminosilicate framework. The cationic charge of the metal or hydrogen ion balances the negative charge on the aluminosilicate framework.
Aluminosilicates are formed by polymerization of SiO4 and AlO4 tetrahedra to form sheet like polyhedral. The polyhedra forms cubes, hexagonal prisms and truncated octahedral. These 3D tertiary building blocks in turn are arranged regularly to form a superstructure inside which pores and supercage exists. Each supercage is characterized by a window size aperture which can block entry of sufficiently large molecules. This is known as sieve effect. The zeolite structures have pores oriented in one, two or three directions leading to 1D, 2D, 3D structures. Structures of zeolite X and A is shown in Fig 1.
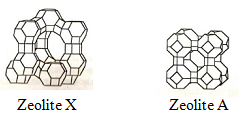
Fig 1. Schematic diagram of zeolites structures
Zeolites are classified based on their pore diameter and ring size. Among aluminosilicate zeolites 3A, 4A, 5A and erionite containing 8 number of rings have pore diameters in the range of 3-5 Å. ZSM 5 and mordenite with 10 and 8 rings respectively have pore diameters in range of 3-5 Å. On the other hand, faujasite X andY have 12 rings with larger pore diameter of 7 - 8 Å. Aluminophosphates (ALPOs) have significantly extended range of pore sizes. ALPOs containing 12 rings have pore diameter of 10 Å while 18 ring ALPOs have pore diameter of 10-15 Å.
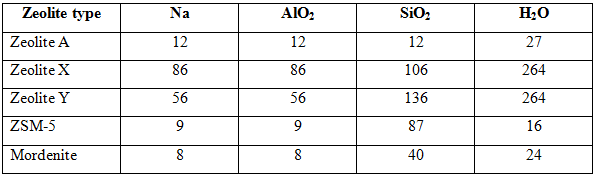
Table 1. Compositions of common zeolites per unit cell