Catalyst tests
Activity and selectivity
The performance of catalyst is characterized by its activity for reaction and selectivity for a product. The activity of a catalyst for a reaction at specified conditions is generally expressed in terms of rate of reaction at that condition over the catalyst. The catalyst that shows higher rate of reaction at the given conditions is said to have higher activity. For solid catalyzed reaction, rate can be defined as, r = g mol of reacted /s. g of catalyst. Here mass of solid is used because amount of catalyst is important. The reactor volume that contains the catalysts is of secondary importance. Activity is also expressed in terms of conversion of a reactant achieved. Higher the conversion at given conditions, higher is the activity of the catalyst for that reaction. The conversion can be defined as follows
![]()
For supported metal catalysts, defining activity in terms of metal sites is more useful as the metal sites actually act as the active sites for chemical reactions. Hence, activity is often expressed in terms of turnover frequency or TOF. TOF is defined as the number of molecules reacting per active site per second (s-1). Though the TOF value is expected to be constant for a particular metal catalyst for a given reaction at specified conditions, however, in reality significant difference may be observed due to differences in catalyst preparation, metal support interaction, crystallite size, surface structure and morphology etc.
![]()
The next important parameter determining the performance of catalyst is selectivity for a particular product. The selectivity of a product X can be defined in several ways. One of the definitions is shown below:
![]()
The performance of catalyst is tested in a suitable experimental setup. Fig. 2 shows a schematic diagram of a simple experimental setup for studying gas phase reaction over solid catalysts. Typically, solid catalysts are tested in a tubular down-flow reactor. A simple manometer or any pressure gauge can be included before the reactor to ensure that the pressure drop is within acceptable limits. All the reactants are combined in a mixer and sent to the reactor where it comes in contact with the catalyst bed. The product gases or liquids are usually analyzed using a gas chromatograph equipped with suitable columns. The columns are selected so that the product compounds are separated efficiently giving distinct peaks that can be easily identified and quantified. Further detector type and temperature as well as carrier gas type and flow rates are also important parameters determining the retention time and peak area in a chromatograph. Catalytic tests are carried out at predetermined range of temperature, pressure, feed composition and total flow rates depending on reactions. The catalysts amount range to be used is also fixed according to the requirement.
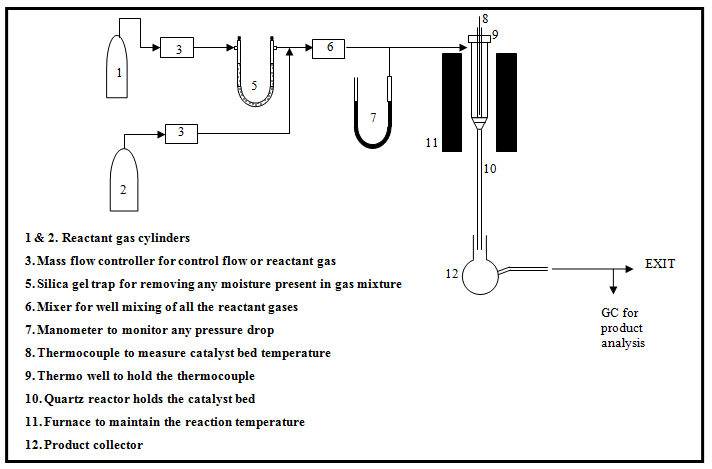
Fig. 2. Schematic diagram of a typical experimental setup
2.3.2.1. Collecting data from laboratory reactors
Kinetic data are collected for the following purposes:
• Determination of activity-selectivity and deactivation data for catalyst selection
• Determination of reaction mechanism and kinetic parameters for understanding the reaction at the fundamental level so the reaction process can be modeled which can be used for design of reactors.
Investigation of the reaction kinetics is done at steady state conditions for most active and selective catalyst. The effect of temperature and partial pressures of reactants on the activity and selectivity is investigated in the desired temperature range. The process of data collection typically involve three major steps:
- Selection of a reaction and catalyst
- Selection of reactor type
- Analysis of data
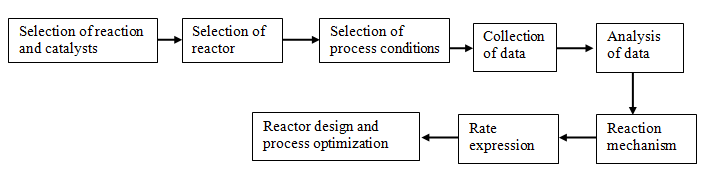
Fig. 2. Steps for obtaining and analyzing kinetic data
To measure the specific catalytic activity or intrinsic reaction kinetics, the data must be collected in absence of any pore diffusion limitations, mass transfer limitations or heat transfer limitations. For analysis of data collected in the presence of mass transfer or heat transfer limitations, suitable criterion should be included during analysis. Deactivation of catalysts should be also avoided during data collection. These will be discussed in detail in later sections. Further data should be collected over a wide range of temperature and reactant concentrations to provide adequate reaction model that can be applicable at wide temperature and concentration range.
Mode of reactor operations
Reactors can be operated in two ways:
- Integral reactors
- Differential reactors
Integral reactors
When a reactor is operated at high conversion, the reactor is said to be an integral reactor. When reaction takes place in a packed bed reactor at high conversion there is possibility of wide variation of conversion and reaction rate within the reactor even under isothermal conditions. Hence, the integral form of design equation is to be used. This is the basis for design for an ideal packed bed reactor.
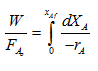 |
(1) |
Differential reactors
When the reaction in a tubular reactor is carried out at very low conversions the changes in conversion and reaction rate across the reactor are small enough to be neglected and the rate can be considered constant across the reactor. Then the rate in above equation [1] can be considered constant and taken out of the integral and the equation can be simplified to
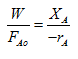 |
This above equation is same as for mixed flow reactor or CSTR. In this case analysis of data is simplified.
Book Reference
• J. M. Smith, Chemical Engineering Kinetics, McGrawHill Book Company, 1981
• J.J. Carberry , Chemical and catalytic reaction Engineering, Dover Publications,2001
• D.W.Green, R.H. Perry, Perry´s chemical engineer´s handbook, 8 th Edition, McGrawHill Company,2007
• H. S. Fogler, Elements of Chemical reaction engineering, Prentice Hall of India, 1999
• R. J. Farrauto, C. H. Bartholomew, Fundamentals of Industrial catalytic Processes, Blackie Academic & Professional, 1997