Multiphase reactors
Slurry reactors
The catalytic reaction can also be carried out in two–phase or three –phase stirred tank reactors also known as slurry reactors. In three –phase reactor, gas and liquid reactants are brought into contact with solid catalyst particles. In two–phase reactor, fluid phase is usually liquid reactant in contact with the solid catalyst. The reaction of gaseous reactant with catalyst is usually carried out in fixed bed reactor. In three –phase slurry reactor the gaseous reactant and solid catalysts are dispersed in continuous liquid phase by mechanical agitation using stirrer. The efficient stirring ensures nearly uniform composition throughout the reactor. This kind of reactor is used in hydrogenation, oxidation, halogenations and fermentation process. The advantages include nearly isothermal operation and good heat and mass transfers. The use of powder catalysts having high activity minimizes the intraparticle diffusion limitation. The reactors can be operated in batch, semi batch or continuous mode. In three – phase system bubbles of gas rise through agitated slurry. Solid particles are in size range of 0.01to 1.0 mm. The solid concentration can be up to 30 vol. %. Lower concentration is also used. In hydrogenation of oil with nickel catalyst, the solid content is 0.5 vol. %. The external transport effects are important in slurry reactors and details are discussed in lecture no. 30. Hydrogenation of oils is carried out in slurry of nickel catalyst particles. Industrial hydrogenation reactors are usually of the size in the range of 500-200 L. The reactors are operated up to pressure of 200 atm and temperature of 350°C. The reactors are equipped with internal agitator, gas inlet, facility for insitu sampling and heater or cooler for temperature control.
Trickled bed reactors
In trickled bed reactor gaseous and liquid reactants flow co-currently downward over a packed bed of solid catalyst particles. The liquid is distributed across the reactor cross section by a distributor plate. The gas enters at the top and distributed along with the liquid. The liquid flows downward by gravity and drag of the gas. For low liquid flow rates and low to moderate gas flow rates, the gas phase is continuous with liquid trickling down forming film over the solid catalyst. The thickness of the liquid film has been estimated to vary between 0.01 and 0.2 mm. This flow regime is known as a trickle flow regime. The fluid approaches plug flow leading to higher conversion than slurry reactors for the same reactor volume. Other advantages include ease of installation, minimal catalyst handling and low catalyst attrition as in packed bed reactor. Disadvantages include maldistribution of flow resulting in channeling or bypassing, possibility of non uniformity in packing, incomplete contacting or wetting and intraparticle diffusion resistance. Catalyst bed depth is limited by pressure drop, catalyst crush strength and maximum adiabatic temperature increase for stable operation. The reactor length to diameter ratio can vary between 1 and 10 depending on the allowable pressure drop. Other parameters important for trickled bed include void fraction of bed, holdup for phases, wetting efficiency (fraction of catalyst wetted by liquid), gas – liquid mass transfer coefficient, liquid–solid mass transfer coefficient, liquid and gas mixing, pressure drop and heat transfer coefficients. The wetting efficiency of the catalyst is important for reaction rate and increases with increasing liquid rate. The trickle bed reactor is most commonly used for hydrogenation and hydrodesulfurization reactions.
Bioreactors
In bioreactor live cells or enzymes are used as catalyst to perform the biochemical reactions. Bioreactor operations are limited by the conditions favorable for the biological systems. Most living cells can tolerate only mild conditions of temperature and pH. Hence in bioreactors stringent control of temperature, pH or any contamination is needed. Bioreactor may have two phases, liquid-solid as in anaerobic process or three phases, gas, liquid and solid as in aerobic process. The solid phase typically contains the cells (bacteria, fungi, algae etc.) that serve as biocatalyst. The density of biocatalytic phase is close to water. The biocatalyst can also be used in immobilized form in which cells are trapped within solid or semi solid structure such as porous particles or gel. Liquid is primarily water with dissolves the feed and products. In aerobic bioreactor the gas phase consists of primarily air and product gas CO2. Bioreactors are mainly operated in batch or semi batch mode allowing better control of process parameters. Increasing number of bioreactor is operated in continuous mode such as in wastewater treatment, lactic acid production, production of human insulin etc.
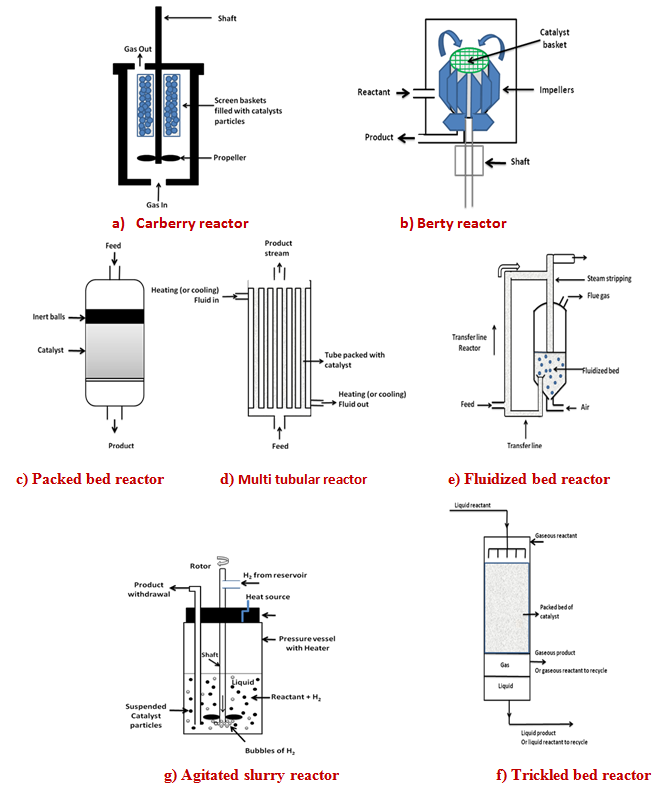
Fig. 1. Schematic diagram of different type of reactors