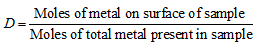Working principle and instruments
In pulse chemisorption technique, initially the sample is reduced with hydrogen at elevated temperature to convert the oxide to metallic form (such as NiO to Ni). Then, the sample is flushed with an inert gas at elevated temperature, such as helium, to remove traces of any adsorbed gases. Then, sample is cooled to the analysis temperature, with inert gas flowing. Successive small pulses of adsorbate gas (of known volume) are injected in the flow of inert gas by a syringe or loop at the desired temperature. Pulses of adsorbate gas are continued to be inject ed until the catalyst surface is saturated. The Fig 2 shows a typical plot of peak area of adsorbate gas as function of number of pulse injected. As the figure shows, the first pulse corresponds to negligible peak area suggesting that most of the injected gas is adsorbed. For second pulse the peak area increased compared to the first suggesting lesser amount of gas adsorbed compared to first injection. As the number of pulse increases peak area gradually increases corresponding to decreased amount of gas being adsorbed. Finally the peak area becomes constant corresponding to the area of pulse volume. The constant areas are shown by pulse number 6 and 7 in the Fig 2. The amount of gas adsorbed is obtained by calculating the difference between the volume of each pulse and the fraction of H2 not adsorbed. The total amount of gas chemisorbed can be obtained from the number of consumed pulses. The schematic diagram of equipment for pulse chemisorption is given in Fig. 3.
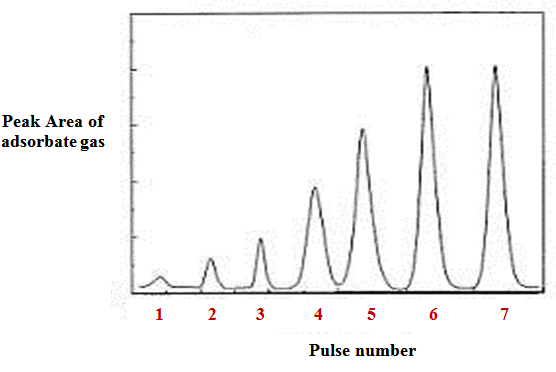
Fig. 2. A typical plot of peak area vs pulse number of adsorbate gas
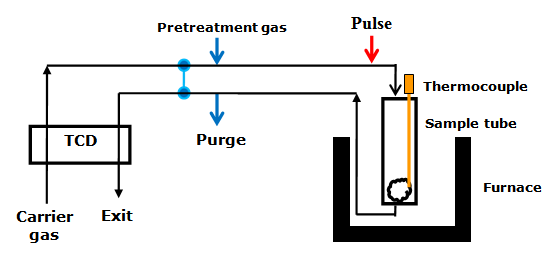
Fig. 3. Schematic diagram of equipment for pulse chemisorption
Calculations for determination of metal dispersion and active metal area:
1. The volume of active gas (CO or H2) injected ,Vinj , can be calculated at STP conditions using the equation below. Generally, the active gas is used as a gas mixture, such as 10% CO in helium or 10 % H2 in argon.
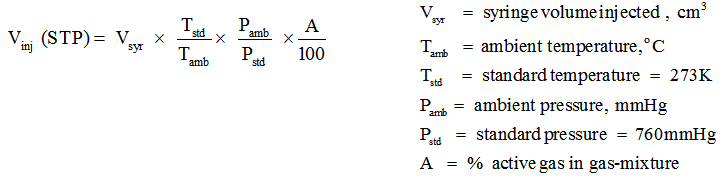
2. The volume of active gas chemisorbed is calculated using volume injected Vinj and from area under the peaks as follows.
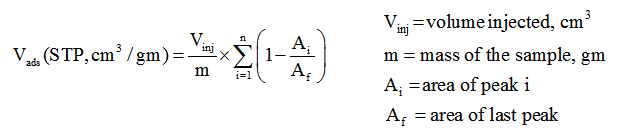
3. Calculation of % Metal Dispersion
Moles of metal on surface of sample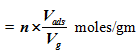
Moles of total metal present in sample ![]()