Chemisorption
In many catalysts active metals are deposited on the surface of the support such as in Pt/Al2O3, Pt/C, Ni/Al2O3 etc. These metals act as active sites in various oxidation states including in zero valence state. The location and state of the active metal on the support largely depend on the preparation method of the catalyst. When prepared by impregnation method, more active metals are expected to concentrate on the surface of support, whereas in bulk preparation method active metals are trapped more within the bulk matrix. Further, these metals usually exist as clusters on the support surface and thereby, only a fraction of the total deposited metal is actually exposed to the incoming reactant molecules for participation in a reaction.
The dispersion (D) of metal is defined as 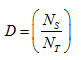
Where ,
Ns = total no. of exposed surface atoms
NT = total no. of metal atoms present in the sample
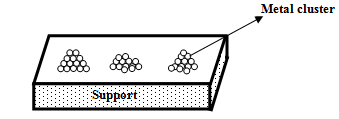
Fig. 1. Metal cluster on support surface
Chemisorption technique gives direct measurement of the number of exposed surface atoms. This method measures quantity of gas adsorbed selectively on the metal at monolayer coverage.
This technique is mainly used to study:
(1) metal dispersion and
(2) active metal area.
In this method, the sample is exposed to a gas that can chemisorb on the active metal. At monolayer coverage, the average number of surface metal atoms associated with adsorption of each gas molecule is determined. Accurate determination of metal dispersion and the active metal area is possible if stoichiometry of the chemisorption reaction is known. Pulse method is frequently used to determine the number of gas molecules adsorbed on the catalyst surface.
For a given catalyst, the adsorbate gas is chosen to minimize adsorption on the support and to have an irreversible (or weakly reversible) chemisorptions on the metal. H2 and CO are the most commonly used adsorbate gases.
Hydrogen Chemisorption
H2 adsorb dissociatively on metals according to the equation:
![]()
Ms represents a surface metal atom.
The chemisorption stoichiometry n is defined as the number of metal atoms to which one adsorbate gas molecule can attach. In case of hydrogen chemisorption one hydrogen molecule attaches with two metal atoms; hence n = 2.
CO chemisorption
The CO adsorption is considerably more complex. The CO can chemisorb in various forms on metals such as Fe, Ni, Ru, Pt, Pd. The chemisorption stoichiometry varies with temperature, metal dispersion, metal loading and preparation. CO can be chemisorbed:
- Dissociatively (CO: n = 2 ) or
- Associatively
- Linear (n =1)
- Bridged (n = 2)
- Multi-bonded species (n = 3)
The relative proportion of the various forms depends on temperature, pressure and metal particle size.
Adsorbate gas choice
For a given catalyst, the adsorbate gas should be chosen such that it interacts irreversibly with metal or have very weak reversible interaction. The adsorbate should have minimum adsorption on the support. Most commonly used adsorbate for metal characterization is hydrogen and CO. Other gases such as O2, N2O, NO, N2 etc. are also used depending on the specific application. The CO can chemisorb in different forms on metals such as iron, nickel, ruthenium, palladium, rhodium and platinum. Not knowing the exact stoichiometric relation can introduce error. The main difficulty is the formation of metal carbonyl, which are volatile as in case of nickel and rhodium. However, CO chemisorption is better suited when hydrogen can be absorbed into the metal (palladium hydride formation) or significantly adsorbed on support (such as carbon support). Oxygen chemisorption is also used. However, possibility of formation of metal oxides with variable stoichiometry may cause errors. Main application of oxygen adsorption measurement is hydrogen-oxygen titration. For cases, when interaction of metals with H2 or CO is very low and interaction with oxygen is excessively strong resulting in bulk oxidation, such as for Cu or Ag, the adsorptive decomposition of nitrous oxide can be used.