Procedure:
1. J774A.1 cells are cultured in the DMEM media containing 10% FBS and 1% antibiotics cocktails (pencillin/streptomycin sulphate).
2. Remove the cells from the cell culture plate by trypsinization or by 0.5% EDTA in PBS.
3. Plate 10,000 cells on 12mm cover glasses and incubate it in the 24 well dish with 0.5ml DMEM media containing FBS and antibiotic cocktail.
4. Incubate cells over night at 370C and 5% CO2 and it will allow the cells to attach to the cover glasses.
5. Wash the cells with DMEM without FBS media.
6. Prepare a suspension of latex beads (106 beads/ml) in DMEM without FBS media.
7. Remove media and add beads suspension to the well and centrifuge the 24 well dish at 1000rpm for 1mins at 40C.
8. Incubate the plate for 1hrs at 370C and 5% CO2.
9. Wash the well with 1ml DMEM without FBS media to remove unentrenalized beads.
10. Fix the biological sample with Methanol: Acetone (7:3) mixture at -200C for 15 min. Hydrate the sample with 1X PBS.
11. Stain the cells with filipin (50µg/ml) for 1hrs at 370C in dark.
12. Keep one drop (~20µl) of mounting medium (glycerol mounting media containing antifading agent) on the glass slide and keep the cover glass on it. Firm the cover glass by making a thick rim by nail polish.
Observation: Observe the cells in the bright field and look for the beads on the cells. Observe the cells in the fluorescence microscope with UV filter.
Results : A typical phagocytosis of bead will represent by the appearance of beads in the phase and the same bead will be circled by fluorescence (Figure 33.1).
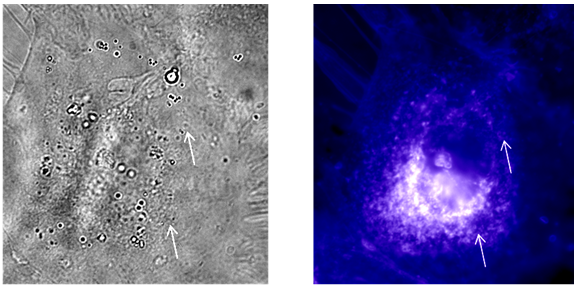
Figure 33.1 Observation of macrophages fed with latex beads after staining with filipin. Arrow indicates the position of phagocytosed beads.