2. Preparation of TNF-α Standard dilution: Dissolve the vial content into 1ml deionized water to yield a stock standard 30ng/ml. Allow the standard to equilibrate for at least 15 minutes before making dilutions. For preparing different concentration of TNF-α solution, initially prepare a 1000 pg/mL standard from the stock standard. Vortex to mix. Dilute this stock into different dilutions as per the calculation given in the Table 30.2. and follow as given in the Figure 30.5
Table 30.2: Preparation of the TNF-α dilutions |
|||
TNF-α concentration (pg/ml) |
TNF-α (µl) |
Assay Dilution Buffer (ml) |
Total volume |
1000 |
300 |
300 |
600 |
500 |
300 |
300 |
600 |
250 |
300 |
300 |
600 |
125 |
300 |
300 |
600 |
62.5 |
300 |
300 |
600 |
31.25 |
300 |
300 |
600 |
15.62 |
300 |
300 |
600 |
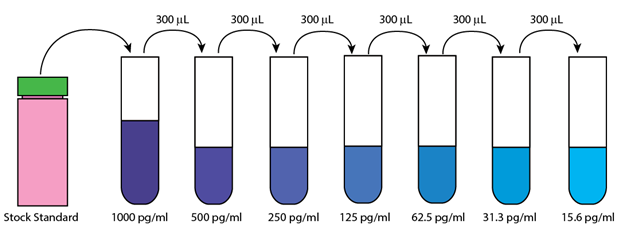
Figure 30.5: Procedure to prepare the serial dilution of TNF-α
3. ELISA plate coating: Add 100 µL diluted Capture Aantibody to each well. Incubate overnight at 4°C. Aspirate and wash 3 times with wash buffer.
4. Blocking : Add 200 µL Assay Diluent to each well. Incubate 1 hr RT. Aspirate and wash 3 times with wash buffer.
5. Add 100 µL standard TNF-α or sample to each well. Incubate it for 2 hr at RT. Aspirate the sample and wash the plate 5 times with wash buffer.
6. Detection: Add 100 µL Working Detector (Detection Ab + SAv-HRP) to each well. Incubate for 1hr at RT. Aspirate the detector solution and wash 7 times with wsh buffer. Add 100 µL Substrate Solution to each well and incubate 30 min RT in dark. Stop the reaction by adding 50 µL Stop Solution to each well. Read the ELISA plate at 450 nm and the at 570 nm (it is required to substract background absorbance).