Although can be used with any type of chromatographic method, the major separation modes of HPLC are:
- 1. Size exclusion chromatography (discussed in lectures 22 and 23)
2. Ion exchange chromatography (discussed in lecture 24)
3. Normal phase and adsorption chromatography: In principle, this mode is the column version of thin layer chromatography. The stationary phase is polar (such as silica gel, with or without functionalization with the polar groups) while the mobile phase is non-polar.
4. Reversed-phase chromatography: The name ‘reversed-phase' has historical significance. In 1970s, liquid chromatography was usually performed with polar stationary phases, usually unmodified silica or alumina. Polar molecules will preferentially bind to the polar stationary phase. This method is now termed as the normal phase chromatography. If the stationary phase is made hydrophobic through covalent attachment of long alkyl chains, it would preferentially bind the hydrophobic molecules. This can be considered the opposite or reverse of the normal phase, hence the term reversed-phase chromatography.
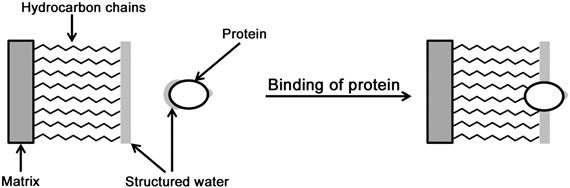
Reversed-phase chromatography finds applications in both analytical and preparative biochemical separations. Since its introduction, reversed-phase chromatography has come a long way to become the method of choice for analytical purposes. The solute molecules that possess some hydrophobic character bind to the hydrophobic stationary phase; reversed-phase chromatography, therefore, is a type of adsorption chromatography. The actual mechanism of binding of the molecules is not very clear but is believed to happen due to favorable entropic effect. The initial mobile phase in reversed-phase chromatography is primarily aqueous. Binding of the solute's hydrophobic regions to the stationary phase reduces the exposed hydrophobic area thereby diminishing the degree of ordered water structure and increasing the entropy of the system (Figure 26.2). The bound analyte molecules can then be eluted using a gradient of a non-polar solvent.
|
Figure 26.2: Interaction of a protein to a reversed-phase column; binding of hydrophobic regions of the protein to the hydrophobic stationary phase would displace the structured water molecules thereby increasing the entropy of the system. |