Q 4: You are provided with acetic acid (density: 1.05 g/cm3) and sodium acetate (CH3COONa). Calculate the amount of the acetic acid and sodium acetate required for preparing a 200 ml of 250 mM buffer of pH 5.0. Marks=5
Q 5: The table shown below shows the initial reaction rates of an enzyme in th presence of different substrate concentrations.
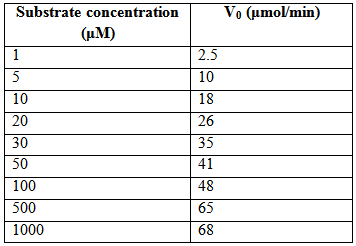
Draw Lineweaver-Burk plot and determine the Km and Vmax for the enzyme. Marks=5
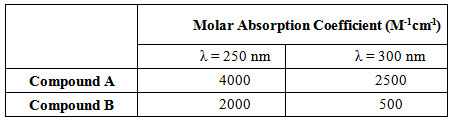
Q 6: You are given a solution having two components, A and B. The absorbance values of the solution at 250 and 300 nm were found to be 0.3 and 0.25, respectively. Calculate the concentrations of the two components in the sample using the data given in the table.
Marks=5
Q 7: The C=O stretching vibration band for acetone appears at 1700 cm-1 in an infrared spectrum.
- Calculate the frequency of carbonyl stretching vibration in Hz (vibrations/second). Marks=2
- Will the N–H stretching vibrational frequency for dimethylamine be higher or lower than that of C=O stretching vibrational frequency? Marks=1
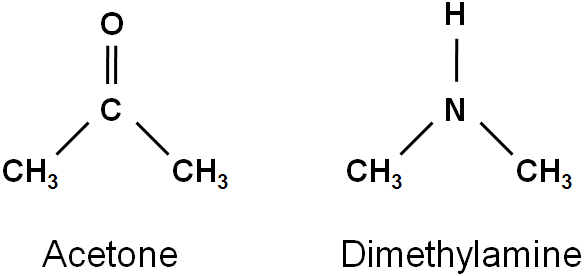
- Explain the reasoning behind your answer in (b). Marks=2
Q 8: Explain the purpose of following chemical reagents for PAGE. Marks=10
- Acrylamide
- SDS
- NN methylene Bis-Acrylamide
- TEMED
- Coomassie Blue
- Bromophenol Blue
- Ammonium per sulphate
- 2-mercapto ethanol
- Tris-HCl
- Glycine