Oligomeric status of the protein- Native molecular weight determination by gel filtration in conjugation with the SDS-PAGE can be used to determine the oligomeric status of the protein.
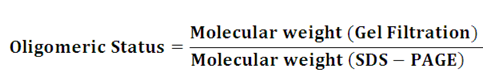
Studying protein folding- Protein is madeup of the different types of amio acid residues linked by the peptide bond. As soon as peptide chain comes out from the ribosome, it folds into the 3-D conformation directed by the amino acid sequence, external environment and other factors. Protein structure has multilevel organization; Primary structure (sequence of protein), secondary (α-helix, β-sheet and turn), tertiary and quaternary structure. When protein is incubated with the increasing concentration of denaturating agents (such as urea), it unfolds the native structure into the unfolded extended conformation following multiple stages. The different protein conformation forms during unfolding pathway has distinct hydrodynamic surface area and it can be use to follow protein folding-unfolding stages with the gel filtration chromatography. The details of the experimental setup is given in the Figure 33.1. Protein is incubated with different concentration of urea (0-8M) for 8-10hrs at 37°C. A gel filtration column is equiliberated with the buffer containing urea (same as in incubation mixture) and the incubation mixture is analyzed. As the concentration of denaturating agent is increasing, protein will unfold with an increase in hydrodynamic surface area. As a result, protein peak shifts towards left. At highest concentration of denaturant, protein unfolds completely and mostly appear in void volume.
Studying protein-ligand interaction- Gel filtration chromatography separates the molecules based on their size. Ligand binding to the protein induces conformational changes, result into the change in size or shape (Figure 33.2). In addition, ligand is small in size where as protein-ligand complex is big and may appear at a distinct place in the column. In step 1, a gel filtration column is equiliberated with the buffer and elution profile of ligand is recorded. Now column is equliberated with the buffer containing ligand molecule. As the concentration of ligand is increased, protein binds ligand and form a larger complex with an increase in hydrodynamic surface area. As a result, protein peak shifts towards left. As the concentration of ligand will