Features of major biomolecules
Classification of biomolecules is largely based on their chemistry. There are four major classes of biomolecules: proteins, nucleic acids, carbohydrates, and lipids.
Amino acids and proteins
Proteins constitute the functional machinery in the living systems by carrying out most of the biological reactions. They are the unbranched polymers of L-α-amino acids. D-amino acids do exist in nature, but such molecules are rare. The structure of a typical amino acid is shown in Figure 1.3A.
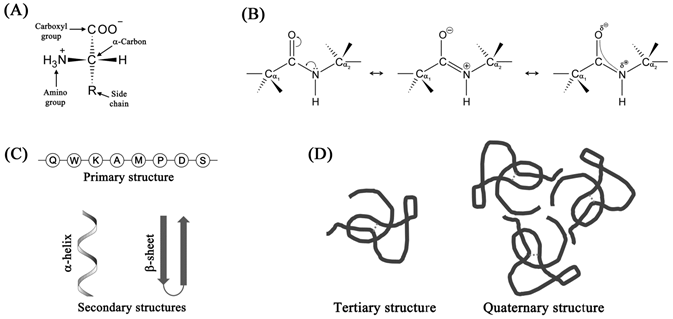 Figure 1.3 Structures of amino acids and proteins: structure of a typical L-α-amino acid (A); peptide bond showing the partial double bond character (B); primary and secondary structures (C); and tertiary and quaternary structures (D) formed by proteins.
Figure 1.3 Structures of amino acids and proteins: structure of a typical L-α-amino acid (A); peptide bond showing the partial double bond character (B); primary and secondary structures (C); and tertiary and quaternary structures (D) formed by proteins.
The R group (shown in Figure 1.3A) is what differentiates the 20 standard amino acids present in proteins. During protein synthesis, the amino acids are linked together through an amide bond, called peptide bond (Figure 1.3B). Delocalization of nitrogen's lone pair of electrons over carbonyl group imparts a partial double bond character to the peptide bond putting severe conformational constraints on the polypeptide backbone (Figure 1.3B). The sequence of amino acids in a polypeptide chain is termed as its primary structure (Figure 1.3C). The linear polypeptide chain can adopt local higher order structures stabilized through hydrogen bonds; these local ordered structures are termed secondary structures (Figure 1.3C). Two such secondary structures found in proteins are: α-helices and β-sheets. Further folding of the unstructured regions in the polypeptide chain results in a compact structure, termed the tertiary structure, the highest structural level of a single chain protein (Figure 1.3D). Certain proteins function as multimers wherein more than one polypeptide chains assemble together through non-covalent interactions to form what is called a quaternary structure (Figure 1.3D).