29.6 Gene Therapy for Cystic Fibrosis
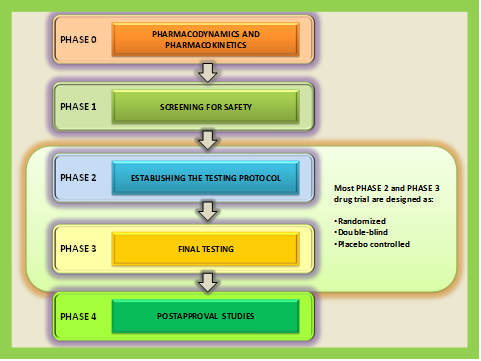
Drug trial has been categorized into five phases as shown in the flowchart of drug trial phases below (figure 29.3).
The phase 2 and most phase 3 drug trials are currently designed as randomized, double-blind, and placebo-controlled.
Randomized: In the randomized design each study subject is randomly assigned to receive either the study treatment or a placebo (fake treatment).
Blind: In this form of study targeted individuals do not know about the treatment. In case of double-blind study the clinician also do not know the kind of treatment being given to the targeted individuals. The purpose of 'blinding' is to prevent biasness, since if a clinician knew about the treatment regime, he/she might be tempted to manipulate the study. Moreover, a clinician might be baised in sampling and analyzing the data in order to show the higher efficacy of the tested gene therapy product.
There is another form of double-blind design known as the “double-dummy” design which provides additional security against biasness or placebo effect. In this design the patient is administered with both placebo as well as active doses alternatively during the study period.
Placebo-controlled: The placebo helps the researchers to differentiate the effect of the study treatment from the placebo effect and effectively analyze the case.
Figure 29.3 Different phases of drug trial: