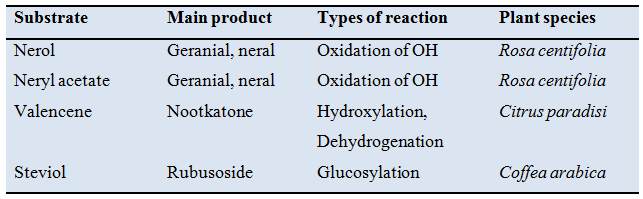
Table 38.1: Examples of biotransformation
3. Biotransformation using immobilized cell culture
Entire cells offer the chance to implement multistep biotransformation and to utilize and recycle essential cofactors and co-enzymes. Isolated enzymes may be sensitive to denaturing conditions, including pH, heat and specific organic solvents. In order to be useful in biotransformation reactions, biocatalysts need to be stable and reusable. Use of whole cell immobilized system may help overcome some stability problems. Immobilized plant cells have some additional advantages over freely suspended cells. They are more resistant to shear damage and can be used repeatedly over a prolonged period. Complete cell immobilization may also create adverse conditions under which secondary metabolite production may be improved. A very common method for immobilization of plant cells are gel entrapment by ion exchange, precipitation, polymerization and in preformed structures. For adsorption of plant cells, solid surfaces can be used. Enzymes may be adsorbed to insoluble supports by hydrogen bonding, dipole–dipole interactions and hydrophobic interactions. Commonly used supports are polypropylene and diatomaceous celite.
4. Genetic engineering approaches towards biotransformation
Bioconversion/biotransformation capacity of cell cultures can be further improved by changing the following parameters:
- Cell selection
- Elicitation
- Permeabilization
- Radiation
- pH of medium
- Osmotic shock
A more basic approach is the transfer of genes that code for the key enzymes catalyzing the desired biosynthetic reactions into a fungal or bacterial cell because of their ability to produce high amounts of enzymes (Pras et al., 1995). Hashimoto et al. (1993) explain the expression of hyoscyamine 6-b-hydroxylase in Escherichia coli . This recombinant bacterium was capable to convert hyoscyamine to scopolamine. Subsequently, this cloned gene has been transferred to Atropa belladonna and expressed constitutively. Additionally, he also found that transformed hairy roots with increased efficiency of conversion of hyoscyamine to scopolamine. Cloning and expression of bacterial lysine decarboxylase under the control of a 35S promoter fused to the coding sequences of the small subunit of rubisco transit peptide in tobacco root cultures was found to affect two secondary metabolic pathways (Berlin et al., 1998).