5.4. Shear sensitivity
Sensitivity of plant cells to hydrodynamic stress related with aeration and agitation can be ascribed to the physical characteristics of the suspended cells, their size, the presence of thick cellulose based cell wall, and presence of large vacuoles. Aeration and mixing system, aeration rate and impeller tip speed generally decide the shear-related effects on plant cells. Mechanically agitated vessels may over-aerate plant cultures, in addition to damaging and breaking the cells through the hydrodynamic stress generated by aeration, agitation, shaking, pumping, and other operations. Low agitation and high aeration provide oxygen in a reasonable mixing range.
5.5. Optimization of process parameter
Nutrients directly influence the yield and productivity of metabolites in plant cell suspension cultures. Therefore, it is important to study and quantify the effect of selected key medium components on growth as well as product accumulation and strike a balance between the two to enhance the yield and productivity. This is essential for secondary metabolites production as conditions suitable for growth may adversely affect the product formation and vice versa. The first step in bioprocess media optimization is the identification of relatively significant media components, such as sugars, nitrogen compounds, minerals and growth factors as well as culture conditions and then to determine their optimum levels. The growth of cells in the bioreactor is controlled by using concentration of the growth-limiting nutrient. At steady-state the cell density and substrate concentration are constant. At steady state, μ = D , where D = F / V ( F = medium flow rate, V = culture volume).
6. Types of bioreactors
In vitro plant cell culture is currently carried in a diverse range of bioreactor designs, ranging from batch, airlift, and stirred tank to perfusion and continuous flow systems. For a small-scale operation, both the conventional and novel bioreactor designs are relatively easy to operate. For a larger scale of operation, problems of maintaining bioreactor sterility and providing adequate oxygen supply to the cells have yet to be resolved. The bioreactors used for plant cell cultures are classified as under:
- Mechanically agitated bioreactors: stirred tank reactor equipped with various propellers (spin, helix, bladed, paddle), rotary drum tank reactor, etc.
- Air driven bioreactors: bubble column, concentric tube airlift reactor, external loop airlift reactor, propeller loop reactor, jet loop reactor, etc.
- Non-agitated bioreactors: (a) packed bed, (b) fluidized bed, (c) membrane reactor.
Three important scientific and practical issues are involved in bioreactor design and operation for plant cells:
- Cell growth and product formation assessment
- Modeling of the culture dynamics, including the integration of biosynthesis and product separation
- Studies involving the flow, mixing and mass transfer between the phases, in order to define criteria for bioreactor design and scale up
6.1. Mechanically agitated bioreactors
The various plant bioreactors designs are proposed by various authors depending upon the plant species used (see Bisaria et at, 2002). The most common and popular bioreactor is the stirred tank bioreactor and sufficient knowledge exists about its design and applications. Although it has gained much popularity, stirred tank bioreactors have numerous limitations, such as high power consumption, high shear, and problems with sealing and stability of shafts in tall bioreactors. In order to diminish the shear forces, numerous modifications have been developed by employing a variety of impeller designs and seals (Figure 36.2.Aa).
Horizontal vessels or rotary drum reactors (Figure 36.2.Ab) have significantly higher surface area to volume ratio than other reactor types. Therefore, mass transfer is achieved with comparably less power consumption. Horizontal vessels used for the cultivation of high-density plant suspensions have shown advantages in terms of suspension homogeneity, low shear environment and reduced wall growth, over either airlift or stirred tank reactors. However, the drawback is their comparatively high energy consumption in large scale operations.
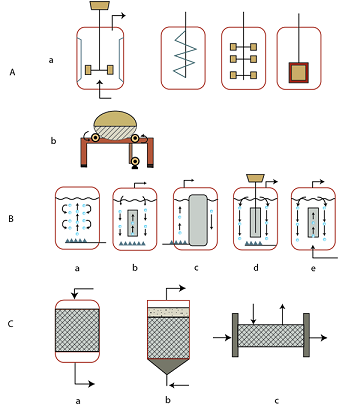
Figure 36.2: Bioreactor types for plant cell, tissue and organ cultures: (A) Mechanically agitated bioreactors: (a) stirred tank reactor equipped with various propellers (spin, helix, bladed, paddle), (b) rotary drum tank reactor; (B) Air driven bioreactors: (a) bubble column, (b) concentric tube airlift reactor (IL ALR), (c) external loop airlift reactor (EL ALR), (d) propeller loop reactor, (e) jet loop reactor; (C) Non-agitated bioreactors: (a) packed bed, (b) fluidized bed, (c) membrane reactor.