1. Introduction
Plant tissue culture is the key method of propagation for a large number of commercially important plants including important vegetatively propagated crops. Over 600 millions plants can be produced in a year by tissue culture methods. Agar culture is the main culture technique generally used for commercial tissue culture propagation. It requires a large number of small culture vessels and labor, and results in the requirement of many laminar-air-flow clean benches, large autoclaves, and large culture spaces equipped with illuminated shelves, electric energy, etc. Thus, it is the cause for both limited propagation efficiency and high production costs. In order to overcome these problems, many attempts for establishing large-scale production of propagules with simple production facilities and techniques have been made including robotics, photoautotrophic cultures, bioreactor techniques, etc. Bioreactor technique seems to be the most promising technique among them in reducing the labour, and providing low production cost, which will be sufficient for establishing a practical system for in vitro mass propagation and commercialization of plants. A bioreactor may be referred to as any manufactured or engineered device that supports a biologically active environment. Bioreactors are widely used for industrial production of microbial, animal and plant metabolites as by allowing large-scale cultivation of cells. A simple diagram of a bioreactor is shown in Figure 36.1.
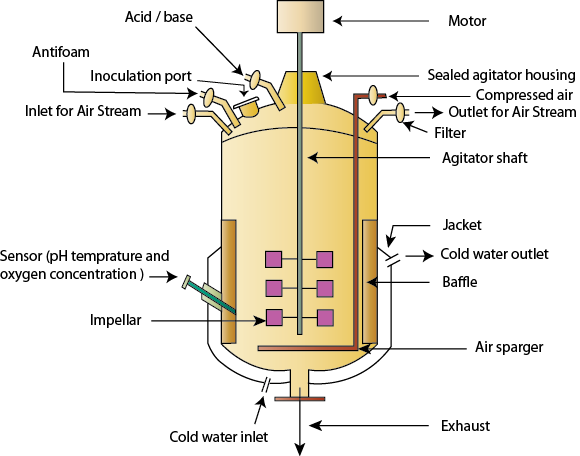
Figure 36.1: Diagram of a typical bioreactor .
2. Process format
The choice of culture system influences the productivity. There are three main methods for the mass culture of plant cells, i. batch culture system, ii. continuous culture system and iii. semi-continuous (draw-fill). Out of these , the most studies on plant cells have been based on batch cultures system grown in fermentors (or bioreactors) (Fig.36.1). I n batch cultivation , an inoculum of known cell density is “seeded” into a specified volume of preconditioned medium in the bioreactor and allowed to grow for a definite period under controlled conditions. The cells containing the desired intracellular metabolite are harvested from the fermentor at the end of the culture period. Typically , nothing is added or removed from the bioreactor during the course of cultivation, except addition of acid/base to control the pH and air to supply oxygen to respiring plant cells. To start a new process, the bioreactor is cleaned, sterilized and filled with sterile medium and inoculated with the desired plant cells . Various environmental parameters such as pH, temperature and dissolved oxygen can be controlled with the use of pH, temperature and dissolved oxygen probes and the corresponding control systems. In batch culture, the t ime taken for cleaning etc can be considerable but can be eliminated by the use of continuous culture. In a continuous system, the nutrients consumed by the tissues are continuously replenished by an inflow of fresh medium to the bioreactor. A constant inflow of fresh medium is maintained by a constant efflux of equivalent volume of spent medium plus cells. However, continuous culture of plant cells is difficult because of the presence of aggregates which give a non-homogeneous culture and make sampling difficult. The adhesion of the cells to the walls of the bioreactor also gives problems. The slow growth of the culture means that the supply of fresh medium has to be at a slow rate, which in practice, is difficult. One method of avoiding continuous culture is ‘ draw-fill' or ‘semi-continuous ' culture where, at the end of the culture period, 90% of the culture is removed and the remaining 10% topped up with fresh, sterile medium. This avoids the cleaning and sterilizing the vessels between the runs.
The continuous or draw-fill cultures are suitable for the production of secondary products that are ‘growth related'. However, secondary products are generally non-growth related and accumulate only after growth has ceased. To improve or stimulate secondary product yield, the medium or culture conditions are often changed; thus, continuous culture or draw-fill are not suitable. The non-growth related accumulation requires a two-stage process, which can be organized by using a batch culture. In such a system, in the first stage growth of plant cells is optimized and the cells are transferred to a second stage. The second stage contains the nutrients in which product formation takes place in the cells. The culture conditions in the second stage are normally different from those in the first stage.