1. Fractionation
Fractionation is a separation method in which a certain quantity of a mixture (solid, liquid, solute, suspension or isotope) is divided into number of smaller quantities (fractions) in which the composition changes according to a gradient. Fractions are collected based on differences in a specific property of the individual components. Fractionation process makes it possible to separate more than two components in a mixture in a single run. It is generally carried out by suspending each extract in water separately and partitioning with different organic solvents, such as hexane, chloroform, ethyl acetate, and methanol in order of increasing polarity by using separating funnel. A simple fractionation unit is shown in Figure 34.1. All the fractions of plant extract can be dried by evaporating respective solvent using rotary evaporator and can be stored at 4°C till further analysis.
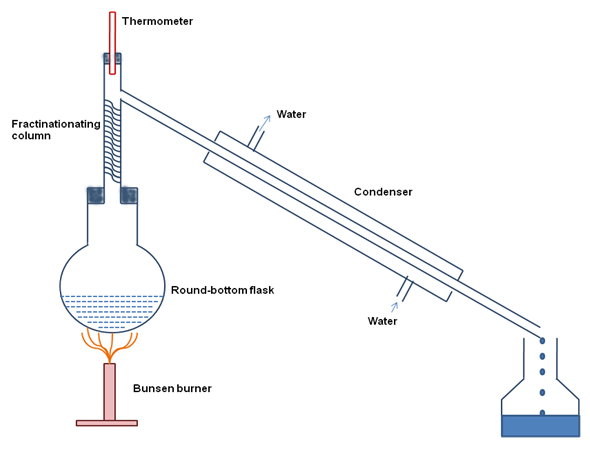
Figure 34.1: A simple fraction distillation apparatus
Qualitative phytochemical analysis can be done for various phytoconstituents, like alkaloids, tannins, glycosides, steroids and saponins, by thin layer chromatography (TLC) of obtained fractions in different solvent system. High performance liquid chromatography (HPLC) is an important device for quantitative analysis of phytochemicals, their monitoring and quality assurance. The method enables complex mixtures to be separated into individual compounds, which can be identified and quantified by suitable detectors and data handling systems. Separation and detection occurs at ambient temperature or slightly above. Therefore, the method is preferably suited for compounds of limited thermal stability.
2. Bioassays
Bioassays are generally conducted to measure the potency of a substance by its effect on living cells and are useful in the development of new drugs. Biological assays must be carried out in order to identify plant extracts, to guide the separation and isolation, and to evaluate lead compounds. These assays may be conducted both in vitro or in vivo .
2.1. Antiviral Assays
A number of compounds extracted from different species of higher plants have shown antiviral activity. Examples included tannins, flavones, alkaloids that displayed in vitro activity against numerous viruses. Antiviral assays are basically an extension of cytotoxicity assays. To perform this, the cultures of mammalian cells are infected with virus, test compounds are added, and the fate of the cells is assessed. It was reported that the antiviral activity detected was due to phytoalexins produced by the plant as a protection against plant viruses. These assays simultaneously allow an estimation of cytotoxicity (loss of the cell monolayer in which the plaques are normally formed). Active extracts or compounds then become candidates for testing against tumor cell lines, for example, P388 or L1210.
The methanol extracts of the aerial parts of Hypericum mysorense and Hypericum hookerianum , exhibited detectable antiviral effect towards HSV-1 with an inhibitory concentration at IC50 value of 50µg/ml. The acetone extract of Usnea complanata also showed antiviral activity at an IC50 value of 100µg/ml. Other examples of plant extracts exhibiting anti-HSV activity are Acnistus arborescens, Cupania glabra, Dichapetalum axillar, Drypetes lasiogyna, Mallotus mollissimus.
The pentacyclic triterpenoids, betulinic acid, oleanolic acid and ursolic acid, are widespread plant metabolites. All the three triterpenes inhibit HIV-1 protease activity in vitro. Betulinic acid was found to be active in vivo as well using athymic mice carrying human melanomas. Further biological studies suggest that betulinic acid works by induction of apoptosis.