Lately, biologically and highly active recombinant human insulin-like growth factor-I (rhIGF-I) was produced in yeast (S. cerevisiae). rhIGF-I is a 7.5kDa protein containing 70 amino acid residues, which stimulates the proliferation of a wide range of cell types including muscle, bone and cartilage tissue. IGFs control the biosynthesis of many intracellular and extracellular components and are potent mitogens for MDCs (mesenchymally derived cells). Growth hormone has been shown to mediate its effects on bone formation indirectly, through IGF-I. rhIGF-I is also known as Somatomedin-C is effective and its use apparently involved no special hazards.
Recombinant DNA technology is mainly used as a key for production of the growth hormone. In 1979, Dr. Baxter's team and a group of scientists at Genentech succeeded in producing human growth hormone in genetically modified bacteria. Recently, these artificially derived biosynthetic growth hormones are widely exploited in humans.
7-3.4 Production of Monoclonal Antibodies (mAb) Using Microorganisms as Cell Factories:
Monoclonal antibodies are specific antibodies which bind to the particular site of proteins i.e. epitope. Production of the monoclonal antibodies was done from the identical immune cells. They had a major role in treatment of cancer due to their site specificity.
Major techniques involved in the production of recombinant monoclonal antibodies were repertoire cloning or phage display/yeast display. Recombinant antibody techniques use viruses and yeast as a cell factories for the production of monoclonal antibodies.
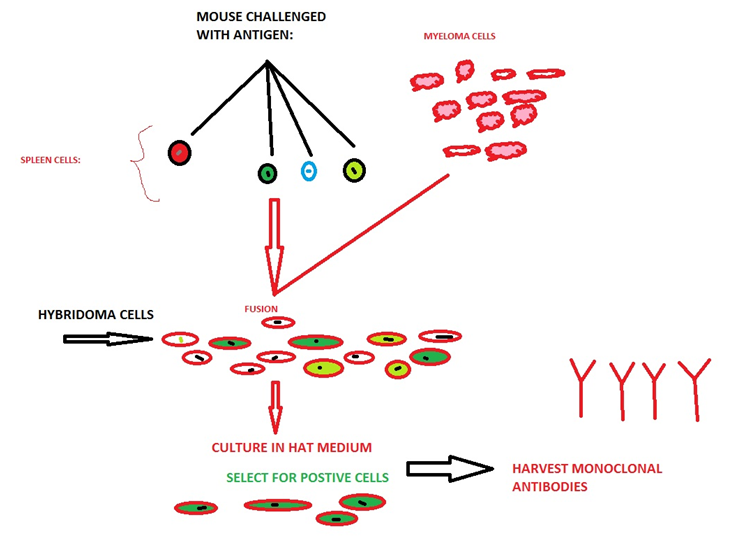
Figure7-3.4: Conventional protocol for production of monoclonal antibodies