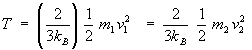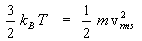 ................................... ( 15 ) ................................... ( 15 )
Thus the thermal energy of an ideal gas is equivalent to the average kinetic energy per molecule.
Let us consider a mixture of two gases at thermal equilibrium, then according to eqn(15),
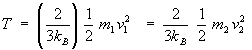
or 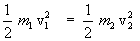 ............................... ( 16 ) ............................... ( 16 )
Here 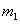 and and 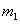 are the moleculer mass of each gas and are the moleculer mass of each gas and 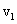 and and 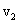 are the rms velocity of molecule of each gas. are the rms velocity of molecule of each gas.
According to eqn(16), at thermal equilibrium the Kinetic energy of different kind of gases is same. Thus the heavier molecule moves slower than the lighter molecules, since 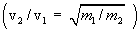 . . |