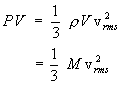4.Kinetic Energy and Temperature From the basic laws of the the thermodynamics, we know that the change in temperature of the system is associated with the change in internal energy. Thus higher the temperature of the system, larger the internal energy. We can show that the internal energy is related to kinetic energy and there is a direct relation between kinetic energy and temperature of a gas as described below. The pressure exerted by the gas on the wall of the cubic container is reproduced below, |