Electron Spin Resonance Spectroscopy:
For example, if we look at the EPR spectrum of radical anion of benzene as shown in Figure 36.05, we see seven lines with different intensities. This is due to fact that the electron is delocalized over all six carbon atoms and therefore exhibits coupling to six equivalent hydrogen atoms.
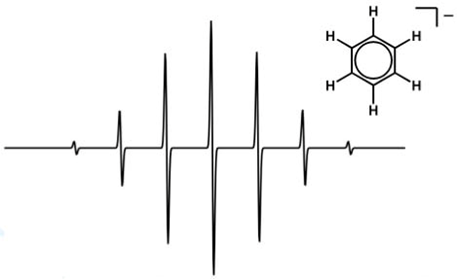
Figure 36.05: EPR Spectrum of benzene radical anion [1].
No useful information can be obtained for paramagnetic compounds from NMR spectroscopy, but EPR spectra can provide additional insight. For instance, the analysis of the coupling patterns can provide information about the number and type of nuclei coupled to the electrons. The magnitude of A can indicate the extent to which the unpaired electrons are delocalized and g-factors can show whether unpaired electrons are based on transition metal atoms or on the adjacent ligands.
Ref.[1]. http://joule.qfa.uam.es/epr/tutorial_uk/5_1_5Benzene_anion_radical_.html.
Quiz 36:
(Q36.1) What is EPR or ESR spectroscopy?
(Q36.2) Why do we need high frequency in the range of GHz for EPR study?
(Q36.3) Can we study EPR spectrum without applying magnetic field? Why?
(Q36.4) What is called hyperfine interaction? How can we understand from EPR spectrum?
(Q36.5) Can you observe EPR spectrum for typical antiferromagnetic materials?
(Q36.6) How does the EPR spectrum of simple ferromagnetic materials look like?