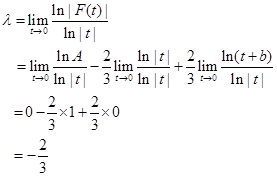
However, ![]() and
and ![]() both have λ = o as per the definition given in Eq.2.1. The first function has logarithmic singularity whereas the second one has cusp like singularity. The above definition thus can not distinguish these two singularities. A modified definition can be adopted for the critical exponent to distinguish such singularities.
both have λ = o as per the definition given in Eq.2.1. The first function has logarithmic singularity whereas the second one has cusp like singularity. The above definition thus can not distinguish these two singularities. A modified definition can be adopted for the critical exponent to distinguish such singularities.
If for a smallest integer j , the jth derivative of the function, ![]() , diverges as
, diverges as ![]() , the exponent will be given by
, the exponent will be given by
...........................................................![]() ..........................................(3.2)
..........................................(3.2)
Using this definition, let us find the exponent λ that describes the logarithmic singularity in ![]() and the cusp like singularity in
and the cusp like singularity in ![]() .
.
1. ![]() :
:
![]()
Thus, ![]() .
.
2. ![]() :
:
![]()
Thus, ![]() .
.
Therefore, the definition (2.2) can distinguish the logarithmic and cusp like singularity.
One then can calculate all the critical exponents associated with the thermodynamic quantities defined in section 2, in the limit ![]() following either Eq. (3.1) or (3.2).
following either Eq. (3.1) or (3.2).
However, one may wonder why critical exponents are so important when they contain less information than the complete function. Firstly, in experiments, sufficiently close to T c the behaviour of the leading term in the thermodynamic quantities ![]() dominates and they can be described as
dominates and they can be described as ![]() . Therefore it is easy to estimate the critical exponents but the full function may not be. Secondly, there exists a large number of relations among the critical exponents and they are not all independent. In fact, only two of them are independent (which will be shown later). Thus, determining only two exponents one may obtain the values of rest of the exponents.
. Therefore it is easy to estimate the critical exponents but the full function may not be. Secondly, there exists a large number of relations among the critical exponents and they are not all independent. In fact, only two of them are independent (which will be shown later). Thus, determining only two exponents one may obtain the values of rest of the exponents.