| |
Heat of combustion of methane
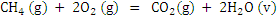 |
(13) |
| CH4 (g) + 2O2 (g) = CO2 (g) + 2H2O (l) |
(14) |
Heat of combustion = (Heat of formation of products) − (Heat of formation of reactants)
By substituting the values of heats of formation, one obtains heat of combustion equals
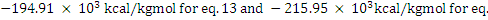 . 14 respectively. . 14 respectively.
Similarly, combustion equations for 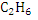 and and 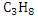 can be written and heat of combustion value can be calculated. can be written and heat of combustion value can be calculated.
Heat of combustion of 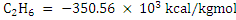 and of and of 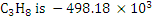 kcal/kgmol when reference state of POC is vapour. Thus net calorific value of natural gas is kcal/kgmol when reference state of POC is vapour. Thus net calorific value of natural gas is
|NCV| = 0.94 ×194.91 × 103 + 0.03 × 350.56 × 103 + 0.005 × 498.18 ×103
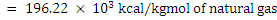
= 8.76 × 103 kcal/m3 (1 atm and 273K)
References:
(1) R. Schuhmann: Metallurgical Engineering, Vol.1 Engineering Principles
(2) O.P.Gupta: elements of fuels, furnaces and Refractories, Khanna Publishers
|