Determination of CV of Gaseous Fuel
In determination of gaseous fuel, combustible components are :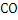 , ,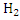 , Hydrocarbons, , Hydrocarbons,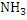 etc. whereas etc. whereas 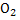 , ,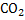 , , 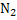 are diluents. are diluents.
Heats of formation of some oxides are:
Oxides |
−ΔHof (1 atm, 298K) in kcal/kgmol |
CO |
29.6 × 103 (C Amorphous) |
CO |
|
CO2 |
97.2 × 103 (C Amorphous) |
CO2 |
94.05 × 103 |
H2O(l) |
68.32 × 103 |
H2O(v) |
57.80 × 103 |
SO2(g) |
70.96 × 103 |
SO3(g) |
94.45 × 103 |
|
Heats of formation of some hydrocarbons are:
Hydrocarbons |
−ΔHof (1 atm, 298K) in kcal/kgmol |
CH4 |
17.89 × 103 |
C2H2 |
|
C2H4 |
−12.5 × 103 |
C2H6 |
20.24 × 103 |
C3H8 |
24.82 × 103 |
|
To note
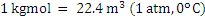
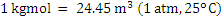
Consider an example of gaseous fuel of the following composition:
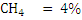
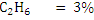
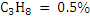
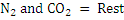
Let us calculate CV of this fuel
In 1 kg mole of gaseous fuel:
|